|
Case Report
Corneal edema from amantadine toxicity in child with fetal alcohol syndrome
1 Associate Professor, Department of Psychiatry, KU Medical Center, Kansas City, KS, USA
2 MD candidate 2025, University of Kansas School of Medicine, Kansas City KS, USA
Address correspondence to:
Ann C Genovese
MD, 3901 Rainbow Blvd, Kansas City, KS 66160,
USA
Message to Corresponding Author
Article ID: 101512Z01AG2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Genovese AC, Longhi S. Corneal edema from amantadine toxicity in child with fetal alcohol syndrome. Int J Case Rep Images 2025;16(2):1–6.ABSTRACT
Amantadine has been used in both neurology and psychiatry for an increasingly wide array of clinical indications, often with significant success in managing difficult-to-treat neurological, developmental, and psychiatric disorders with ongoing research into its potential benefit for many more, often entirely unrelated health conditions. The authors summarize the literature to date which describes corneal edema as a less well-known potential adverse effect of this medication, particularly in children and adolescents. We present a case in which the timely identification of bilateral corneal edema for a child taking amantadine over the course of three years for the treatment of psychiatric and behavioral problems associated with fetal alcohol syndrome, who has resolution of corneal edema following amantadine discontinuation.
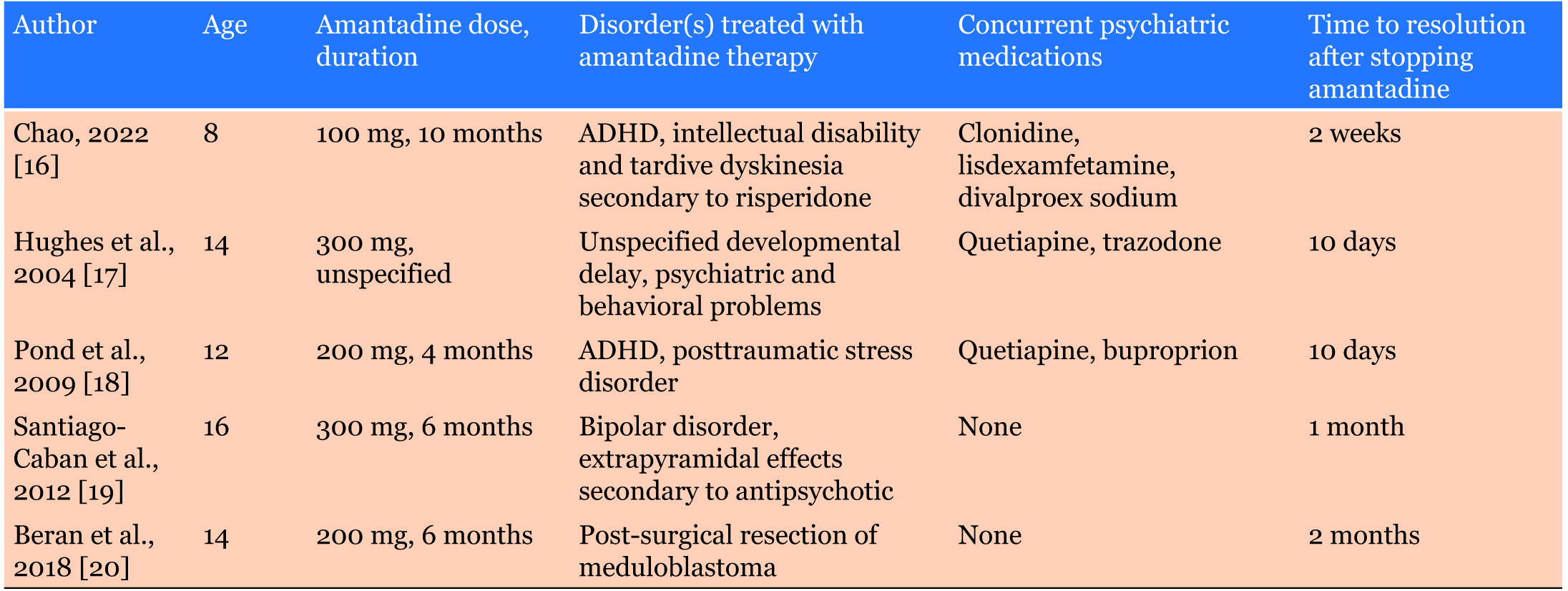
Keywords: Amantadine toxicity, Corneal edema, Fetal alcohol syndrome
Introduction
The mechanism of action of amantadine is related to its antagonism of the N-methyl-D-aspartate (NMDA) receptor, effectively inhibiting the release of glutamate, thus preventing the toxic effects of excess glutamate, which has been implicated in the pathogenesis of many psychiatric disorders and neurodegenerative diseases [1]. Amantadine has been shown to provide neuroprotective benefit by the following mechanisms: (1) anti-inflammatory properties, mainly by inhibiting the release of proinflammatory cytokines; (2) increasing the level of glial cell-derived neurotropic factor; and (3) inhibiting NMDA receptors [2]. Amantadine’s pharmacological actions are unique in combining dopaminergic and glutamatergic properties, which account for its benefit for both Parkinsonian symptoms and levodopa-induced dyskinesias, in addition to less well-defined pharmacological effects, including anticholinergic and serotonergic activity [3],[4].
Amantadine was initially developed for its antiviral properties, demonstrated to have anti-influenza A activity in 1966 [5]. Then, a woman with Parkinson’s disease observed significant improvement of symptoms after taking amantadine for antiviral purposes, following which a clinical study on 163 patients resulted in a positive outcome in reducing tremors and dyskinesias, improving muscle control and lessening rigidity [6]. In the five decades since Amantadine was U.S. Food and Drug Administration (FDA) approved for Parkinson’s disease, amantadine has been investigated for use in various additional indications, including traumatic brain injury, spinal cord injury, multiple sclerosis, Huntington’s disease, catatonia, cutaneous pain, cancer, diabetes, obesity, cocaine abuse, treatment resistant depression, obsessive compulsive disorder, schizophrenia, and drug-induced extrapyramidal symptoms including tardive dyskinesia [4].
Amantadine is often prescribed off-label for behavioral and psychiatric disorders, reported to be generally well-tolerated in children, is considered to have an acceptable side effect profile, and assumed safe for long-term administration [7]. Two randomized controlled trials of amantadine were conducted in children and adolescents, with one reporting reduction of irritability and hyperactivity in autism spectrum disorder at a mean dose of 100 mg BID (twice daily) for one month, and the other showing reduction in symptoms of attention deficit hyperactivity disorder (ADHD) at a mean dose between 100 and 150 mg once daily [8],[9]. In addition, one open-label trial of amantadine for impulsive and aggressive behaviors in eight hospitalized children (4–12 years of age) with developmental disabilities, refractory to other treatments, reported improvement in all eight, with marked clinical improvement in four [10]. The so-called Matthews Protocol was first introduced in 2017, where clinical studies demonstrated that a combination of oxcarbazepine and amantadine significantly lowered the risk of re-hospitalization in children with disruptive mood dysregulation disorder (DMDD) who had been hospitalized for severe irritability and recurrent outbursts [11].
Amantadine-induced corneal edema is a rare but significant adverse drug reaction that was first documented in a 1990 case report for which the therapeutic indication was influenza A [12]. The phenomenon was not more deeply revisited in scholarly work until well into the 2000s, as amantadine gradually increased in use as a treatment for Parkinson’s disease and later for other off-label indications. Currently, amantadine-induced corneal edema has been identified and published in at least 33 other instances, most of which constitute individual case reports, mainly involving adult patients [13].
Of note is a 2010 cross-sectional study involving 169 patients taking amantadine for Parkinson’s disease, in which outcomes of central corneal thickness and endothelial cell density were measured, ultimately concluding that amantadine is more likely to have a negative impact on corneal endothelial cells in a dose-dependent manner when used long term [14]. Most published case reports assert that corneal edema resolves completely upon amantadine discontinuation, however a few cases demonstrate irreversible corneal edema, particularly when the corneal edema was prolonged [15],[16].
Among the existing literature on amantadine-induced corneal edema, only five pediatric cases have been published, all of which describe recovery after discontinuing amantadine [16],[17],[18],[19],[20] (Table 1).
Case Report
Eva is a typically delightful, engaging, and energetic 9-year-old African American female who was referred for psychiatric evaluation, with request for a second opinion. Chief complaints were “extreme irritability” with repeated episodes of reactive aggression, prolonged tantrums, low frustration tolerance and poor impulse control. Eva had significant anxiety, dissociative episodes, distractibility, risk-taking behaviors, defiance, resistance to change and difficulty with transitions. She was noted to be easily influenced by others, would often mimic the behaviors of others, exhibited sexual acting-out behaviors, and was overly friendly with strangers, including adults.
Developmental history is significant for biological mother with intellectual disability who used alcohol during pregnancy, noted to have poor nutrition, lack of prenatal care, and was unhoused. Eva was born by C-Section prematurely at 32 weeks due to placental abruption, with a birth weight of 3.2 lbs. She was adopted immediately after birth, discharged from the hospital after a brief stay and thereafter cared for in the home of her adoptive parents, with whom she developed solid bonding with secure attachment. Pediatric well-child visits demonstrated that Eva was achieving developmental milestones as expected, accounting for prematurity.
Medical history included diagnoses of fetal alcohol syndrome based on dysmorphic features, KDM5B gene mutation with genetic deletion of 2q14.2-q14.3 (including 447 kb), premature adrenarche, wheat/gluten intolerance, psoriasis, low vitamin D level, iron deficiency, chronic constipation, and insomnia. Pharmacogenomic testing shows P450 CYP2D6 and CYP2C19 intermediate metabolizer status, and serotonin 2A/2C receptor HTR2C genotype which is associated with a decreased response to selective serotonin reuptake inhibitors.
Psychiatric history includes diagnoses of separation anxiety, attention deficit hyperactivity disorder (ADHD) inattentive type, fetal alcohol syndrome (FAS) and borderline intellectual functioning. She had tried and failed multiple medications (with reason for discontinuation) of mixed amphetamine salts, atomoxetine, clonidine, topiramate, trazodone, aripiprazole, risperidone (all with behavioral worsening), methylphenidate (psychosis), escitalopram (ineffective), duloxetine (hives), and quetiapine (swelling of tongue and face). Haloperidol with benztropine to prevent extrapyramidal symptoms was helpful but discontinued due to concern of potential long term side effects. Finally, Venlafaxine ER (extended release) was selected due to pharmacogenomic testing results, providing modest but short-lived benefit for anxiety and irritability.
The Matthews Protocol—a combined regimen of oxcarbazepine and amantadine, an off-label regimen originally demonstrated to treat extreme irritability associated with DMDD—was then requested by Eva’s mother after she heard about its purported benefit in a fetal alcohol syndrome support group. Following consultation with a clinical pharmacist, oxcarbazepine was prescribed at 150 mg BID, then amantadine was added at 25 mg BID and eventually titrated to 100 mg BID. This regimen provided significant benefit with improvements in mood, anxiety, dissociative episodes, irritability, tantrums, anxiety, attention span and learning.
However, episodic aggressive outbursts continued to recur episodically over the course of the next 2.5 years, resulting in several psychiatric hospitalizations. Incremental dose increases of oxcarbazepine up to 1500 mg (given in 3 times daily divided dose) and amantadine up to 200 mg BID, which led to improvement in moods and behaviors, but typically lasting only a few months. Finally, guanfacine ER was added, helping to improve sleep as well as alleviating a chronic motor tic of the eyelid.
Neurological symptoms began to emerge at age 12, about 3 years after starting the oxcarbazepine and amantadine regimen, starting with the sudden onset of dramatic, large-amplitude myoclonic jerks of both upper and lower extremities which waxed and waned in intensity, both at rest and during sleep. Initially, their pharmacist suspected this might be due to oxcarbazepine, so the dose was lowered to 900 mg daily in divided doses, however motor movements subsequently worsened. Additional new onset concerns reported during this time included dependent edema of the lower extremities, migratory joint pain, ataxia, slurred speech, and weight loss of 10 pounds despite good appetite and caloric intake.
Comprehensive medical workup included rheumatology consultation which ultimately found no identifiable cause for the tremors. Investigations included brain MRI, lumbar puncture, and labs including autoimmune encephalitis panel, complete blood count (CBC), comprehensive metabolic panel (CMP), creatine kinase (CK), c-reactive protein (CRP), Antistreptolysin O (ASO) titer, serum ceruloplasmin, 24-hour urine copper collection, zinc, lead level, and thyroid-stimulating hormone (TSH), the results of which were all unremarkable. Neurology subsequently prescribed gabapentin 100 mg BID, which resulted in a moderate reduction of tremor intensity.
Then came the report that Eva was “losing her vision.” Shortly after discharge home from the pediatric intensive care unit, Eva developed the acute onset of partial vision loss, found on initial ophthalmologic exam to be affecting only the central vision of the left eye. However, within a period of only a few weeks this had progressed to include central vision loss in the right eye, along with now total loss of vision in the left eye. Eva was fortunate to get an expedited referral to an experienced corneal specialist who diagnosed bilateral keratoconus (a disorder characterized by progressive corneal thinning, with onset typically in the teens or early 20s) and corneal edema (a.k.a. corneal hydrops), defined as an increase in corneal thickness due to the accumulation of extracellular fluid in epithelium and stroma resulting in loss of corneal transparency.
It was concluded that the most likely cause of Eva’s corneal edema was amantadine toxicity. It was also noted that corneal endothelial cell density was probably about half normal for age, which they postulated as a potential explanation as a reaction to amantadine. Treatment recommended at that point included hypotonic eye drops 4 times a day to help pull the fluid out of the cornea, followed by corneal collagen cross-linking, a minimally invasive procedure that can be used to treat corneal edema, particularly in cases of keratoconus, by strengthening the cornea and potentially halting disease progression.
After tapering and stopping amantadine over a period of two weeks, Eva began experiencing an acute exacerbation of psychiatric symptoms, once again experiencing dissociative symptoms and now also expressing what was interpreted as paranoid delusions, all of which improved with low dose lorazepam and olanzapine. During this interval it is reported by family that Eva’s vision began to gradually return to normal. Follow-up ophthalmologic examination performed three months after the discontinuation of amantadine showed resolution of the corneal edema, with visual acuity substantially improved from 20/600 to 20/40. This and subsequent examinations continued to note probable bilateral keratoconus, however it could not be determined whether this was idiopathic or medication induced.
Discussion
Eva’s diagnoses of ADHD, fetal alcohol syndrome, and intellectual disability belied significant behavioral issues of extreme irritability with reactive aggression, prolonged tantrums, dissociative states and poor impulse control. Eva was initially prescribed multiple psychotropic medications from various drug classes, most of which caused either behavioral worsening or adverse effects. Once Eva was started on an oxcarbazepine-amantadine combination per the Matthews Protocol, significant benefits were noted, including reduced irritability, fewer tantrum episodes and improved focus and learning. After the amantadine dose was gradually titrated up to 200 mg twice daily, dissociative episodes were eliminated, with much-improved behavioral stability overall. This amantadine dose was maintained for about three years before signs of corneal edema emerged. Fortunately, within three months of amantadine discontinuation, corneal edema had resolved, and visual acuity was restored.
Eva’s case is interesting and unique for several reasons. Compared to other documented pediatric cases of amantadine-induced corneal edema, she took a relatively high dose of amantadine (400 mg daily) for a significantly longer duration (three years). The onset of bilateral vision loss occurred later than one might expect, and it is possible that this can be attributed to pre-existing keratoconus, co-administration with either oxcarbazepine or guanfacine, or some other unique-to-her factor, which differed from previous pediatric cases [21]. It is also notable that Eva’s corneal edema was indeed reversible within a few months of amantadine discontinuation, making the so-theorized “dose-dependent” nature of amantadine-induced corneal edema somewhat less clear.
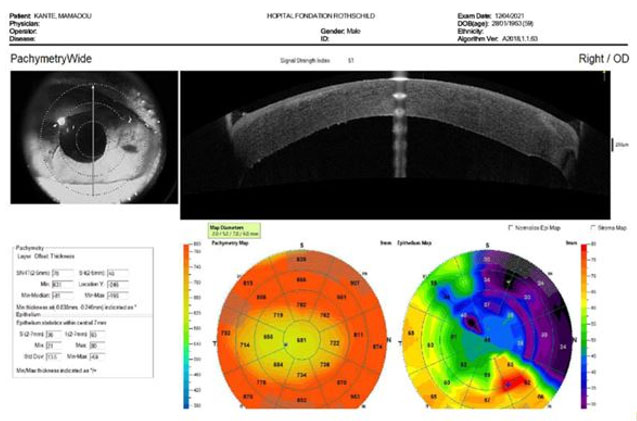
Though Eva’s exam findings were less severe than many cases of corneal edema at the time they are initially identified and diagnosed, her parents were fearful of the worst-case scenario. If corneal edema is severe or untreated it can result in permanent loss of vision or even blindness. Figure 1 shows an example of a corneal optical coherence tomography scan revealing chronic irreversible corneal edema requiring surgical intervention [22].
It should also be noted that Eva reached her historically most optimal level of mood and behavioral stability while taking the oxcarbazepine-amantadine regimen. While returning to this regimen is obviously out of the question for her, the much-enhanced quality of life which Eva experienced while taking it does pose the question as to whether a mirroring treatment with a similar mechanism of action could theoretically approach comparable benefits while avoiding similar serious adverse events.
Conclusion
Children with emotional dysregulation associated with neurodevelopmental conditions including fetal alcohol syndrome present especially challenging cases for family and clinicians alike. In this population it is not unusual for standard treatments to be found ineffective. Thus, it is no wonder that less commonly utilized drugs like amantadine—which represent so much deeper a reach into the child psychiatrist’s toolbox—are being more often considered and prescribed off-label, despite a lower evidence base in comparison to standard treatments and with less well-known adverse effect profiles, particularly in specialized patient populations.
Given the recent emergence of amantadine as a treatment option for pediatric mood dysregulation or irritability, it is imperative that practitioners be cognizant of the risk for corneal edema. Because amantadine toxicity is a rare phenomenon, prescribers may fail to recognize amantadine toxicity and delay drug discontinuation, which in turn reduces the outlook for recovery. Efforts should be made to increase awareness and develop screening and/or monitoring guidelines for corneal edema when prescribing amantadine. With preventative safeguards in place, the demonstrated promise of amantadine as a viable treatment option may not be lost for fear of this potentially debilitating adverse effect.
REFERENCES
1.
Zhou Y, Danbolt NC. Glutamate as a neurotransmitter in the healthy brain. J Neural Transm (Vienna) 2014;121(8):799–817. [CrossRef]
[Pubmed]

2.
Hafizi S, Tabatabaei D, Lai MC. Review of clinical studies targeting inflammatory pathways for individuals with Autism. Front Psychiatry 2019;10:849. [CrossRef]
[Pubmed]

3.
Rascol O, Fabbri M, Poewe W. Amantadine in the treatment of Parkinson’s disease and other movement disorders. Lancet Neurol 2021;20(12):1048–56. [CrossRef]
[Pubmed]

4.
Danysz W, Dekundy A, Scheschonka A, Riederer P. Amantadine: Reappraisal of the timeless diamond-target updates and novel therapeutic potentials. J Neural Transm (Vienna) 2021;128(2):127–69. [CrossRef]
[Pubmed]

5.
Maj J, Sowińska H, Baran L, Sarnek J. Pharmacological effects of 1,3-dimethyl-5-aminoadamantane, a new adamantane derivative. Eur J Pharmacol 1974;26(1):9–14. [CrossRef]
[Pubmed]

6.
Schwab RS, England AC Jr, Poskanzer DC, Young RR. Amantadine in the treatment of Parkinson’s disease. JAMA 1969;208(7):1168–70.
[Pubmed]

7.
Hosenbocus S, Chahal R. Amantadine: A review of use in child and adolescent psychiatry. J Can Acad Child Adolesc Psychiatry 2013;22(1):55–60.
[Pubmed]

8.
King BH, Wright DM, Handen BL, Sikich L, Zimmerman AW, McMahon W, et al. Double-blind, placebo-controlled study of amantadine hydrochloride in the treatment of children with autistic disorder. J Am Acad Child Adolesc Psychiatry 2001;40(6):658–65. [CrossRef]
[Pubmed]

9.
Mohammadi MR, Kazemi MR, Zia E, Rezazadeh SA, Tabrizi M, Akhondzadeh S. Amantadine versus methylphenidate in children and adolescents with attention deficit/hyperactivity disorder: A randomized, double-blind trial. Hum Psychopharmacol 2010;25(7–8):560–5. [CrossRef]
[Pubmed]

10.
King BH, Wright DM, Snape M, Dourish CT. Case series: Amantadine open-label treatment of impulsive and aggressive behavior in hospitalized children with developmental disabilities. J Am Acad Child Adolesc Psychiatry 2001;40(6):654–7. [CrossRef]
[Pubmed]

11.
12.
Blanchard DL. Amantadine caused corneal edema. Cornea 1990;9(2):181. [CrossRef]
[Pubmed]

13.
Raharja A, Mina W, Ashena Z. Amantadine-induced corneal edema: A case and literature review. Am J Ophthalmol Case Rep 2023;32:101881. [CrossRef]
[Pubmed]

14.
Chang KC, Jeong JH, Kim MK, Wee WR, Lee JH, Jeon BS. The effect of amantadine on corneal endothelium in subjects with Parkinson’s disease. Ophthalmology 2010;117(6):1214–9. [CrossRef]
[Pubmed]

15.
Jeng BH, Galor A, Lee MS, Meisler DM, Hollyfield JG, Schoenfield L, et al. Amantadine-associated corneal edema potentially irreversible even after cessation of the medication. Ophthalmology 2008;115(9):1540–4. [CrossRef]
[Pubmed]

16.
Hughes B, Feiz V, Flynn SB, Brodsky MC. Reversible amantadine-induced corneal edema in an adolescent. Cornea 2004;23(8):823–4. [CrossRef]
[Pubmed]

17.
Pond A, Lee MS, Hardten DR, Harrison AR, Krachmer JH. Toxic corneal oedema associated with amantadine use. Br J Ophthalmol 2009;93(3):281, 413. [CrossRef]
[Pubmed]

18.
Santiago-Cabán LA, Rivera E, López-Beauchamp V. Bilateral corneal edema secondary to amantadine in the pediatric population: A case report. Bol Asoc Med P R 2012;104(1):69–76.
[Pubmed]

19.
Beran M, Okyere B, Vova J. Amantadine-induced corneal edema in a pediatric neuro-oncology patient: A case report. PM R 2018;10(10):1122–4. [CrossRef]
[Pubmed]

20.
Chao J, Dunn S, Bohra L. Amantadine-induced bilateral corneal edema in a pediatric patient. J AAPOS 2022;26(3):150–2. [CrossRef]
[Pubmed]

21.
Fan Gaskin JC, Patel DV, McGhee CN. Acute corneal hydrops in keratoconus—New perspectives. Am J Ophthalmol 2014;157(5):921–8. [CrossRef]
[Pubmed]

22.
Ghazal W, Duvillier A, Panthier C, Saad A, Gatinel D. Descemet membrane endothelial keratoplasty with cornea press technique and implantation of a Carlevale scleral-fixated intraocular lens. Am J Ophthalmol Case Rep 2023;32:101882. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Ann C Genovese - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Simon Longhi - Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Ann C Genovese et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.