|
Case Report
Necrotizing and cavitary pneumonia due to Bordetella bronchiseptica transmission from a cat: A case report
1 FRACP, Respiratory Physician, Mackay Base Hospital, Mackay, Australia
2 26th Year Bachelor of Medicine/ Bachelor of Surgery, College of Medicine & Dentistry, James Cook University, Townsville, Australia
Address correspondence to:
Pranav Kumar
FRACP, Respiratory Physician, Mackay Base Hospital, Mackay,
Australia
Message to Corresponding Author
Article ID: 101536Z01PK2026
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Kumar P, Uthayakumar L. Necrotizing and cavitary pneumonia due to Bordetella bronchiseptica transmission from a cat: A case report. Int J Case Rep Images 2026;17(1):38–41.ABSTRACT
Introduction: Bordetella bronchiseptica is an animal-associated respiratory pathogen. Human infection is uncommon but can be severe, especially in older people or those with comorbidities.
Case Report: A 75-year-old woman presented with fever, dyspnea, and productive cough. Imaging showed necrotizing cavitary pneumonia. B. bronchiseptica was isolated on respiratory culture, and close daily exposure to a coughing pet cat was the likely source. She improved with intravenous piperacillin-tazobactam and oral ciprofloxacin, with near-complete radiological resolution at six weeks.
Conclusion: This case highlights the need to consider zoonotic pathogens in atypical pneumonia, to obtain an exposure history, and to target antibiotics based on microbiology and susceptibility results.
Keywords: Bordetella bronchiseptica, Cavitary pneumonia, Necrotizing pneumonia, Zoonosis
Introduction
Bordetella bronchiseptica is a close relative of B. pertussis and B. parapertussis but remains largely an animal pathogen that colonizes the respiratory tracts of dogs, cats, and pigs, producing kennel cough and bronchitis [1],[2]. Human infections are infrequent but increasingly recognized, particularly among older adults and individuals with chronic illness or impaired immunity [3],[4],[5]. Clinical manifestations range from mild bronchitis to severe necrotizing and cavitary pneumonia, empyema, and even systemic disease [6],[7]. Transmission to humans is most often linked to close contact with infected animals or exposure to respiratory secretions and contaminated surfaces [2],[8]. The present case is notable for occurring in a patient with multiple comorbidities but no overt immunosuppression, and for the severity of necrotising cavitary pneumonia.
Case Report
A 75-year-old woman presented with a six-day history of progressive dyspnea, productive cough, and right-sided pleuritic chest chain. She also describes intermittent fevers and fatigue in this period but denies any hemoptysis or recent weight loss.
Past Medical History
Her medical background included type 2 diabetes mellitus, hypertension, hyperlipidemia, asthma, stage 3 chronic kidney disease, and a history of pulmonary embolism managed with long-term anticoagulation. She was a lifelong non-smoker. She reported daily close contact with her pet cat, which had recently developed intermittent coughing.
Examination
On admission, she was febrile (38.2 °C), tachycardic (102 bpm), tachypneic (26 breaths/min), and hypoxemic (oxygen saturation 90% on room air, improving with 2 L oxygen). Her blood pressure was 138/72 mmHg. Chest auscultation revealed coarse inspiratory crackles and reduced breath sounds over the right upper and middle zone.
Investigations
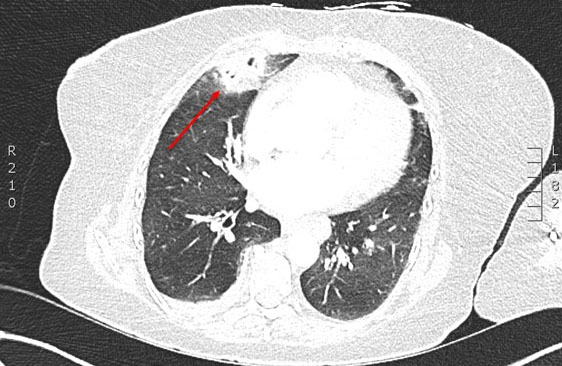
Laboratory results showed a neutrophilic leukocytosis (13.5 × 109/L), a C-reactive protein level of 38 mg/L, and normal liver and renal function. Chest radiograph demonstrated right middle lobe consolidation (Figure 1). Computed tomography (CT) chest revealed necrotising cavitary lesions in the posterior segment of the right middle lobe with surrounding ground-glass opacity and a small pleural effusion (Figure 2). Sputum culture grew B. bronchiseptica, confirmed by standard biochemical testing. Nasopharyngeal polymerase chain reaction (PCR) was positive for rhinovirus. Blood cultures were negative. A good-quality respiratory specimen was collected before antibiotic escalation and processed in the hospital microbiology laboratory using routine methods. Samples were plated onto standard media (including blood and chocolate agar) and incubated at 35–37 °C. B. bronchiseptica was identified using standard biochemical/automated identification, and antimicrobial susceptibility testing was performed using standard methods in line with local laboratory protocols. Contamination was considered unlikely given predominant growth of the organism and concordant clinical and radiological findings.
Management and Outcome
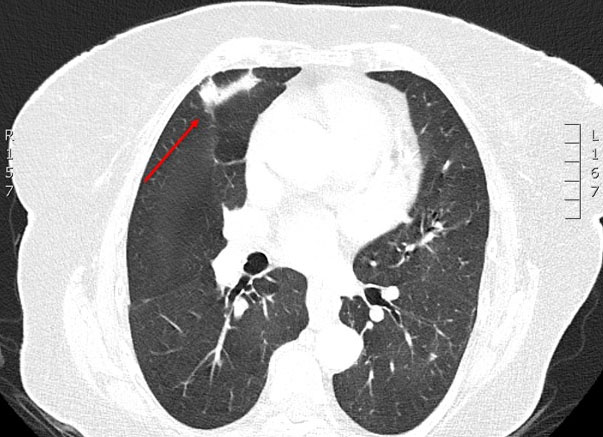
She was started on intravenous ceftriaxone with oral doxycycline for presumed community-acquired pneumonia. After B. bronchiseptica was identified and she continued to deteriorate clinically, treatment was changed to intravenous piperacillin-tazobactam with oral ciprofloxacin. She improved within 72 hours with defervescence and better oxygenation. She completed a 10-day inpatient course, then a short oral course of ciprofloxacin after discharge. At six-week review she was asymptomatic and repeat CT showed near-complete resolution of the cavitary changes (Figure 3).
Discussion
The main reservoirs of B. bronchiseptica are domestic and farm animals [2],[8], which present a source for human transmission through close contact, respiratory droplets, or contaminated fomites. Typically, this organism manifests in immunocompromised patients with predisposing conditions, although cases of immunocompetent patients infection in immunocompetent hosts has also been reported [3],[9].
Human B. bronchiseptica infection may present as tracheobronchitis, necrotizing pneumonia, empyema, or bacteremia [6],[7],[10],[11]. Relapse and persistent airway colonisation have been described, particularly in people with impaired immunity [4],[12]. This case demonstrates that severe necrotizing cavitary pneumonia can occur in older patients with comorbidities. The concurrent rhinovirus detection may have contributed by impairing host defences [11].
Diagnosis requires a high index of suspicion, particularly when there is close pet exposure. Culture and/or PCR can confirm the organism. There is no consensus on radiological or laboratory criteria for diagnosis [5],[13]. There are no formal human treatment guidelines for B. bronchiseptica infection. Therapy is typically guided by susceptibility testing, and agents with good intracellular penetration (for example, fluoroquinolones or tetracyclines) are commonly used [6],[7],[12],[14]. In this case, intravenous piperacillin-tazobactam with an oral switch to ciprofloxacin was associated with a favourable response, consistent with published reports [9],[10].
Conclusion
Bordetella bronchiseptica is a zoonotic respiratory pathogen which can cause severe necrotising and cavitary pneumonia in the elderly or comorbid patients, especially in those with known contact with animals. Workup for this organism necessitates a detailed pet exposure history, and confirmation via microbiological culture is crucial. Antibiotic therapy is determined on a case-by-case basis and depends on the sensitivities and resistances of the isolated organism.
Learning Points
- Zoonotic pathogens such as Bordetella bronchiseptica must be considered when taking detailed patient history, including pet ownership and farm animal exposure.
- Severity of the disease can range from mild respiratory tract infections to severe cavitatory pneumonia, hence necessitating prompt diagnosis with microbiological assays.
- Though there is a lack of consensus on appropriate therapeutics, intravenous (IV) piperacillin-tazobactam and oral ciprofloxacin elicited a favorable response in this case.
REFERENCES
1.
Goodnow RA. Biology of Bordetella bronchiseptica. Microbiol Rev 1980;44(4):722–38. [CrossRef]
[Pubmed]

2.
Mattoo S, Cherry JD. Molecular pathogenesis, epidemiology, and clinical manifestations of respiratory infections due to Bordetella pertussis and other Bordetella subspecies. Clin Microbiol Rev 2005;18(2):326–82. [CrossRef]
[Pubmed]

3.
4.
Woolfrey BF, Moody JA. Human infections associated with Bordetella bronchiseptica. Clin Microbiol Rev 1991;4(3):243–55. [CrossRef]
[Pubmed]

5.
6.
7.
8.
9.
10.
11.
12.
13.
SUPPORTING INFORMATION
Acknowledgments
The authors used an AI language model to assist with grammar and readability during manuscript preparation. All clinical content, interpretations, and the final text were reviewed and approved by the authors.
Author ContributionsPranav Kumar - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Lavieen Uthayakumar - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2026 Pranav Kumar et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.