|
Case Report
A black pleural effusion
1 Department of Respiratory Medicine, Northumbria Healthcare NHS Trust, Northumbria Way, Cramlington NE23 6NZ, UK
Address correspondence to:
Avinash Aujayeb
Department of Respiratory Medicine, Northumbria Healthcare NHS Trust, Northumbria Way, Cramlington NE23 6NZ,
UK
Message to Corresponding Author
Article ID: 101534Z01AA2026
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Aujayeb A. A black pleural effusion. Int J Case Rep Images 2026;17(1):31–34.ABSTRACT
Black pleural effusions are rare, and are associated with cancers, pleural infection (namely fungal), and pancreatic leakage. A 78-year-old with known cardiac comorbidities presented with a large right pleural effusion, and symptoms of breathlessness and weight loss. A computed tomogram (CT) scan showed a large right multiloculated collection and an aspirate of black pleural fluid had negative cytology and grew Klebsiella pneumoniae. Due to the concern that this might represent a malignancy, a thoracoscopy was planned but the patient had to be admitted for cardiovascular instability, and a chest drain was inserted. The local multidisciplinary team (MDT) ruled that this was not a cancer, and a video-assisted thoracoscopic surgery (VATS) was performed for infection clearance. The pleura looked bland, but biopsies were taken, an indwelling pleural catheter (IPC) was inserted. Histology showed a pleural adenocarcinoma and systemic anticancer treatment has been started. Pleural fluid analysis could not be done as it was too dark for routine laboratory analysis which is based on transparency to different wavelengths of light. The cytology was negative, which is not uncommon in patients with lung cancer, and the CT scan was reported as no cancer, again, which is not uncommon. Empyema and concurrent lung cancer is rare. Clinical suspicion was high, and clinicians should always pursue tissue biopsy if that is the case.
Keywords: Black pleural effusion, Empyema, Local anesthetic thoracoscopy, Lung adenocarcinoma
Introduction
Black pleural effusions are unusual. We describe a case where good clinical acumen followed repeated negative investigations to eventually achieve a diagnosis.
Case Report
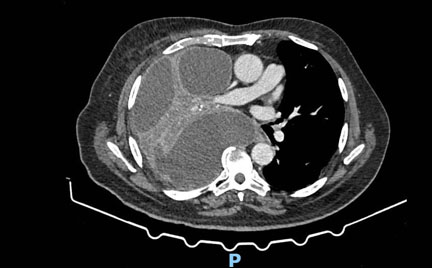
A 78-year-old man, with a past medical history of hypertension, presented to the pleural clinic with a 3-month history of dyspnoea, weight loss, and malaise. His regular medications were amlodipine 10 milligrams (mg) and tramadol 50 mg as and when required. He was an ex smoker of 25 pack years and had probable previous asbestos exposure during his navy work. Blood tests done before the clinic showed haemoglobin levels of 135 (130–180) grams per liter (g/L), white cell count of 13.18 (4–11) × 109 per liter, normal urea and creatinine levels, and a C reactive protein level of 177 (less than 5) milligrams per liter (mg/L). A chest X-ray (CXR) done by the general practitioner had showed a large right sided pleural effusion, and a computed tomogram (CT) scan done urgently, as a two week wait referral, showed a large multiloculated right sided effusion—there was no pleural thickening, masses, or asbestos plaques. Figure 1 is a representative image of the CT scan. An ultrasound showed a large hyperechoic pleural effusion, 2 centimeters (cm) from the skin, and 12 cm deep. Multiple septations and loculations were seen, and there was no discernible pleural thickening, pleural or diaphragmatic masses or nodules. Visual analog score (VAS) for breathlessness was 8. The patient was normotensive, and saturations were 96% on air. Due to the symptoms of the patient, admission was suggested, but the patient refused and instead opted for a large volume therapeutic aspiration. With thoracic ultrasound assistance and with local anaesthetic, 1.5 liters (L) of black fluid was removed aseptically, and as there was a clinical concern of a malignancy as well as of pleural infection, local anesthetic thoracoscopy (LAT) with indwelling pleural catheter (IPC) insertion was planned, and co-amoxiclav 625 mg with amoxicillin 500 mg, both three times a day, were prescribed. Pleural fluid analysis for pH, lactate dehydrogenase, protein and glucose was not possible as the fluid was too dark. Pleural fluid cytology showed predominantly macrophages against a background of red blood cells. There was one cluster of atypical cells noted with conspicuous nucleoli. Cell clot did not highlight any atypical cells and no malignant change was noted. Microbiological growth yielded Klebsiella oxytoca, fully sensitive to co-amoxiclav.
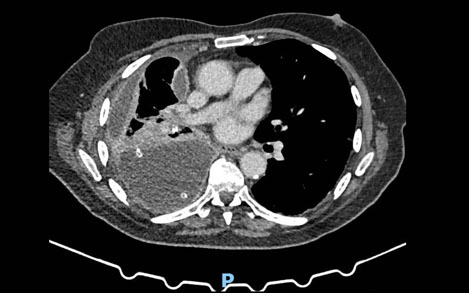
He presented for the LAT and IPC procedure but was in fast atrial fibrillation. The procedure was canceled, and he was admitted for rate control with beta blockers (bisoprolol), and anticoagulation with a direct oral anticoagulant (apixaban). To alleviate this breathlessness, a 12 French gauge Seldinger chest drain was inserted and over 72 hours, 3.1 L of black fluid was drained (Figure 2). Pleurodesis was not performed, and after the patient improved, he was discharged home with an early review in pleural clinic. His antibiotics continued. At that point, five days later, his pleural effusion had re-accumulated. In the interim, he had been discussed at the lung cancer multi-disciplinary team meeting, and it was felt that he had no evidence of cancer, and that this was in keeping with pleural infection. However, a strong clinical suspicion of malignancy remained with the treating physician, and an IPC was inserted to manage the rapidly accumulating fluid. A referral to the regional cardiothoracic centre was made. Video-assisted thoracoscopic surgery (VATS) debridement was undertaken. Septations were broken down and again a few liters of fluid were drained. The pleura looked bland, with no nodular change anywhere, but nevertheless, random pleural biopsies were taken, as is the case for any VATS for pleural disease. The previous IPC was left in situ, and the patient discharged a day later.
The pleural biopsy showed an adenocarcinoma with an immune profile in keeping with pulmonary origin. Contributory factors were his previous smoking history and asbestos exposure. Histological examination showed pieces of pleura with attached adipose tissue. The pleural surface contained epithelioid cells with a papillaroid growth pattern. Similar cells were seen within the pleura and adjacent to adipose tissue, where associated lymphocytic infiltrates were also seen. Immunohistochemistry showed the cells express pan cytokeratin AE1/AE3, BerEP4, monoclonal cancer embryonic antigen (CEA), and strong and diffuse thyroid transcription factor-1 (TTF1). There was no convincing expression of CK5/6 or calretinin by the cells.
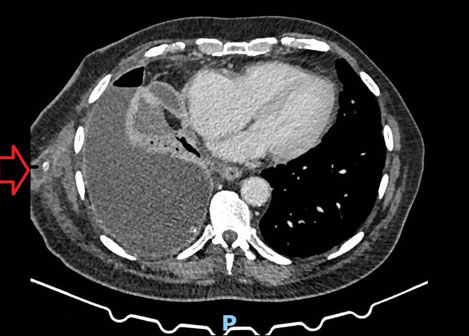
A restaging scan was done (Figure 3) six weeks later, and re-discussion at the lung cancer multidisciplinary team meeting staged the disease at T3N2M1a—the pleural and nodal changes were all felt to be now representative of carcinomatosis. A seroma (Figure 4) was also demonstrated on the CT scan, and the IPC site was leaking—this was successfully with daily drainage of the IPC by the district nurses. Systemic anticancer therapy was started, and the patient continues with active oncological follow-up and the IPC is still in situ, five months later at the time of writing, draining 100 milliliters (mLs) a day.
Discussion
The case above describes a case where high index of suspicion eventually led to a diagnosis of cancer. There are a few notable points for discussion.
First, the initial radiological appearance and the clinical presentation could have all been attributed to a pleural space infection. There was positive microbiological growth from the pleural fluid and prolonged antibiotics with boosted penicillin (to ensure adequate pleural penetration) had been started [1]. However, the weight loss was a red flag (although chronic infection could have caused this as well). The presence of pleural space infection concurrently with malignancy is rarely described in case reports and requires good clinical acumen [2].
The second point is about the color of the effusion which was unusual and alarming. Black pleural effusions are usually attributed to malignancy, pancreatic leaks or specific infections such as with Aspergillus niger or hemolysed blood [3]. There was no issue with the pancreas described on the CT scan, and the aforementioned infection is incredibly rare in a white male Caucasian with no exotic travel history. There was no history of chest trauma. The pleural fluid cytology was also negative and there was no history of melanoma (or one found on thorough skin examination). However, often pleural fluid cytology can be negative in malignant pleural effusions, as the cancer cells might not shed into the pleural fluid, and thus, a negative result can be falsely reassuring. Pleural fluid analysis could also not be done as it was too dark for routine laboratory analysis which is based on transparency to different wavelengths of light [4].
The third point is that there were no appearances on the CT scan or thoracic ultrasound (visceral or parietal pleural thickening, diaphragmatic thickening or nodularity, mediastinal pleural thickening) which was concerning for malignancy [5]. With the above results, the lung cancer multidisciplinary meeting concluded that a cancer diagnosis could not be entertained, and suggested management of the pleural space infection. This is a reasonable approach, although the readership should be reminded that CT scans can be normal in up to 40% of patients with pleural malignancies [6]. Computed tomogram also has low positive predictive value or low sensitivity for concurrent malignancy and empyema [7].
This is the reason why often, access to the pleural via LAT is important to directly visualize any abnormal areas and to enable targeted biopsies [8]. Pleural fluid cytology is also not that helpful in asbestos exposed male patient with exudative effusions, with sensitivity approaching 10% only [9]. The rapid re-accumulation of the pleural effusion, despite a large volume thoracocentesis and a chest drain insertion alongside adequate antibiotics (the CRP had dropped to 88 mg/L after two weeks of treatment) was also a red flag, and as the patient felt well but increasingly breathless, the IPC was inserted as a stop gap measure before definitive surgical therapy. The pleural space was now sterile (repeat cultures were negative). Biopsies are always taken at thoracoscopy (we practice LAT rather than VATS), and multi-site biopsies are the norm, even if the pleura looks normal, as early pleural cancers (for example, mesothelioma in situ) can only thus be detected.
Conclusion
This case has therefore specific learning points:
- Black pleural effusions have several causes, but malignancy needs excluding.
- Negative pleural fluid cytology and a normal CT scan does not exclude malignancy.
- Pleural space infection and malignancy can present concurrently.
- Cytology has low sensitivity in asbestos exposed male patients.
- Despite normal looking pleura at thoracoscopy, multi-site pleural biopsies are crucial to provide a definitive diagnosis.
REFERENCES
1.
Aujayeb A, Conroy K. A case of empyema and a review of practice in a district general hospital. EMJ Respir 2019;7(1):91–96. [CrossRef]

2.
Leporati A, Raveglia F, Cioffi U, De Simone M, Ghelma F, Baisi A. Metastatic lung cancer presenting as thoracic empyema. A case report. Clin Case Rep 2020;8(3):484–6. [CrossRef]
[Pubmed]

3.
Kamal YA. Black pleural effusion: Etiology, diagnosis, and treatment. Indian J Thorac Cardiovasc Surg 2019;35(3):485–92. [CrossRef]
[Pubmed]

5.
Leung AN, Müller NL, Miller RR. CT in differential diagnosis of diffuse pleural disease. AJR Am J Roentgenol 1990;154(3):487–92. [CrossRef]
[Pubmed]

6.
Hallifax RJ, Talwar A, Wrightson JM, Edey A, Gleeson FV. State-of-the-art: Radiological investigation of pleural disease. Respir Med 2017;124:88–99. [CrossRef]
[Pubmed]

7.
Lee HY, Goo JM, Lee HJ, Lee CH, Chun EJ, Im JG. The value of computed tomography for predicting empyema-associated malignancy. J Comput Assist Tomogr 2006;30(3):453–9. [CrossRef]
[Pubmed]

8.
Roberts ME, Rahman NM, Maskell NA, Bibby AC, Blyth KG, Corcoran JP, et al. British Thoracic Society Guideline for pleural disease. Thorax 2023;78(Suppl 3):s1–42. [CrossRef]
[Pubmed]

9.
Arnold DT, De Fonseka D, Perry S, Morley A, Harvey JE, Medford A, et al. Investigating unilateral pleural effusions: The role of cytology. Eur Respir J 2018;52(5):1801254. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Avinash Aujayeb - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthor declares no conflict of interest.
Copyright© 2026 Avinash Aujayeb. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.