|
Case Report
Endocervicosis of the urinary bladder: A case report
1 St. Joseph’s Healthcare Hamilton Charlton Campus, Hamilton, Ontario, Canada
2 Department of Pathology and Molecular Medicine, McMaster University, Hamilton, Ontario, Canada
3 McMaster Institute of Urology, Department of Surgery, McMaster University, Hamilton, Ontario, Canada
Address correspondence to:
Moaz Alowami
St. Joseph’s Healthcare Hamilton Charlton Campus, Hamilton, Ontario,
Canada
Message to Corresponding Author
Article ID: 101528Z01MA2026
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Alowami M, Adelekan O, Liang LMS, Vasudev P. Endocervicosis of the urinary bladder: A case report. Int J Case Rep Images 2026;17(1):1–6.ABSTRACT
Introduction: Bladder endocervicosis (BE) is a rare benign condition characterized by the presence of endocervical-type glands within the bladder wall. Prior Cesarean section or pelvic surgery appears to be a significant risk factor predisposing to BE. Chronic pelvic pain in women is a common symptom of BE. Bladder endocervicosis’s inherent capability to mimic malignancy radiologically and endoscopically may pose a clinical conundrum.
Case Report: A 38-year-old female presented with a five-year history of sharp, stabbing pelvic pain during urination. Magnetic resonance imaging (MRI) revealed a thickened bladder wall and abdominal scar tissue forming a connection between the lower uterine segment and the posterior dome of the urinary bladder. However, no identifiable mass was reported on MRI. Flexible cystoscopy revealed a solid, mass-like lesion with papillary projections on the posterior bladder wall. Histopathological examination of the urinary bladder mass-like lesion revealed benign urothelium with numerous scattered, irregular, and dilated glands lined by columnar mucin-producing epithelium that resembled endocervical glands. Immunohistochemical staining was positive for estrogen receptor (ER) and mucin. Following a 6-week post-operative period, the patient has demonstrated abrupt complete recovery of symptoms.
Conclusion: An effective multidisciplinary approach, including macroscopic evaluation via cystoscopy and histopathological microscopic evaluation, excludes potential malignancy mimickers. Surgical resection leads to complete resolution of symptoms.
Keywords: Bladder endocervicosis, Chronic pelvic pain, Dysuria, Müllerianosis of the urinary bladder
Introduction
Clement and Young were the first to describe bladder endocervicosis (BE) as a rare clinical-pathological dilemma [1]. It is a rare condition characterized by the presence of endocervical-type glands within the bladder wall. Chronic pelvic pain in women is a common symptom of BE. This case’s rare, long-standing history of dysuria compared to more commonly reported pelvic pain in the literature raises awareness for urologists and gynecologists of this atypical presentation [1],[2],[3]. Prior Cesarean section or pelvic surgery appears to be a significant risk factor predisposing to BE. There are two main theories for the pathogenesis of bladder endocervicosis: the implantation theory and the metaplastic theory. This implantation theory suggests that endocervical cells are iatrogenically seeded into the bladder wall during prior pelvic surgeries, such as a Cesarean section. In contrast, the metaplastic theory proposes that multipotent cells derived from the coelomic epithelium undergo hormonally driven transformation into endocervical-type epithelium [1],[2],[3]. Bladder endocervicosis’s ability to mimic malignancy radiologically and endoscopically can precipitate a clinical conundrum. Dissemination of clinical awareness of BE presenting cystoscopically as a solid, mass-like lesion with papillary projections mimicking urothelial carcinoma is critical to mitigate unnecessary aggressive surgical interventions, such as partial or radical cystectomy. This can prevent future harm to the patient due to the potential psychological burden of a cancer misdiagnosis, subsequent high procedural-related morbidity, and diminished quality of life.
Case Report
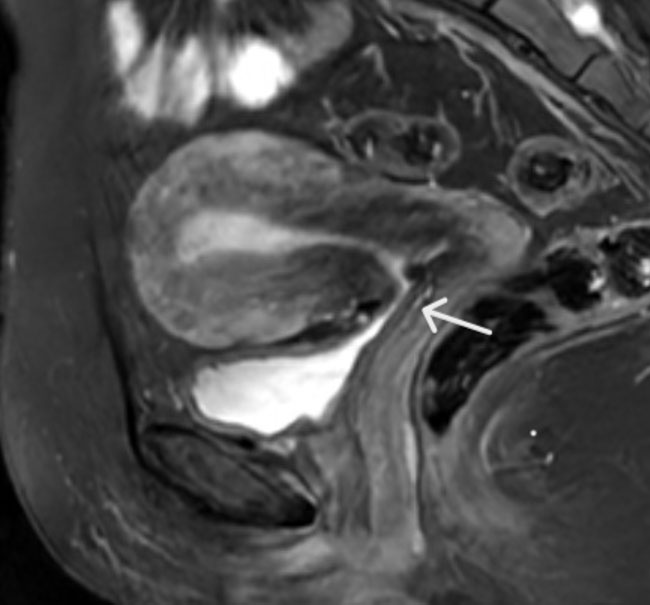
A 38-year-old female presented with chronic, sharp stabbing pain in the lower abdominal area associated with voiding for five years. An unremarkable medical history was reported. Cesarean section preceded the onset of her symptoms. Immediately following a Cesarean section, the patient had urinary retention requiring straight catheterizing for several weeks. She developed pain after voiding almost immediately after surgery. This was described as suprapubic pain radiating to the left hip and down to the leg. The pain was worse during menstruation. No blood was identified in the urine. No systemic symptoms were reported. Complete blood count displayed iron deficiency anemia, and the leucocytes were within normal limits. A magnetic resonance imaging (MRI) scan of the pelvis revealed a thickened bladder wall and scar tissue connecting the lower uterine segment to the posterior dome of the urinary bladder (Figure 1). No discrete intraluminal mass was visualized. Urine cytology was negative for high-grade urothelial carcinoma. No acute inflammation was reported. A urological consultation and flexible cystoscopy were initiated. The cystoscopy identified a solid mass on the posterior wall of the bladder with invaginated papillary projections (Figure 2). The identified mass vindicated potential inflammation, fistula, and scarring. A transurethral resection of the bladder mass was recommended for definitive histopathological assessment. Gross appearance of the mass consists of several fragments of pale-tan soft to firm tissue together measuring 2.5 × 1.7 × 0.4 cm. Microscopic findings of Hematoxylin and Eosin (H&E) stained sections revealed benign urothelium with focal reactive changes, including squamous metaplasia and the presence of numerous scattered, irregular, and dilated glands within the bladder wall (Figure 3). These glands were lined by columnar epithelium with abundant intracytoplasmic mucin, morphologically resembling endocervical glands (Figure 4). There was no associated endometrial-type stroma or evidence of hemorrhage on H&E morphology. No dysplasia or malignancy was identified. The glandular epithelium showed strong nuclear positivity for estrogen receptor (ER) (Figure 5). This case showed classic morphology of endocervical glands with irregular profiles and abundant intracytoplasmic mucin. There was no endometrial stoma seen on the H&E staining, nor were there any features of stomal and glandular hemorrhage that is required for a diagnosis of endometriosis. Further, despite the comprehensive investigations including the MRI, there was no evidence of endometriosis. The surrounding stroma showed some positivity for immunostain CD10, favored to be non-specific staining. Negative CDX2 staining helped exclude intestinal-type metaplasia. Mucin (Figure 6) and Periodic acid-Schiff (PAS) special stains were positive within the endocervical-like glands, confirming their mucinous nature. The pathological findings, notably the presence of endocervical-type glands without endometrial stroma, morphologically with positive mucin stain, strongly favored a diagnosis of bladder endocervicosis.
The patient was subsequently followed up in the clinic six weeks post-surgery and expressed complete resolution of her symptoms.
Discussion
Endocervicosis affects women of reproductive age; the majority are between 30 and 40 years old, like our index case [3]. However, cases in postmenopausal women have been reported [4]. The predominant risk factor for the development of bladder endocervicosis is a history of Cesarean sections or other pelvic surgeries. A review of 40 cases revealed that 75% of patients had undergone prior pelvic surgery [5]. The current literature proposes that the pathogenesis of urinary bladder endocervicosis is explained by two distinct theories. Our case supports the implantation theory, which proposes the iatrogenic seeding of endocervical cells into the bladder wall during pelvic surgeries like Cesarean sections [3]. However, a previous surgical history must exist for this to be a plausible theoretical pathogenesis. In contrast, the metaplastic theory proposes that multipotent cells originating from the coelomic epithelium undergo transformation into endocervical-type epithelium. This hypothesis is supported by the consistent location of lesions on the hormonally sensitive posterior bladder wall, the co-existence of other Müllerian-derived tissues (Müllerianosis), and the strong expression of estrogen and progesterone receptors. The hypothesis is that metaplasia is driven by hormonal differentiation [3].
This case presentation featured a five-year history of chronic dysuria. The suprapubic pain was felt radiating down to the leg and it was worse during menstruation. Clinical presentations in bladder endocervicosis may range from urinary symptoms to pelvic pain. Dysuria is less common in bladder endocervicosis lesions as they often start and remain beneath an intact urothelium, and the urine does not directly contact the inflamed or abnormal tissue. Bladder endocervicosis also lacks the highly inflammatory and hemorrhagic stromal component typical of endometriosis. Most cases of BE in the reported literature present with lower abdominal pain and pelvic tenderness [1]. Differential diagnosis includes gossypiboma, cystitis glandularis, nephrogenic adenoma, and bladder adenocarcinoma (Table 1). Albeit dysuria is a common symptom of BE, when occurring in individuals with a prior history of Cesarean section, it can imitate rare postoperative complications such as gossypiboma. A recently reported case following a previous Cesarean section demonstrated how retained surgical material can mimic common pelvic symptoms and complicate diagnostic interpretation [6].
Bladder endocervicosis’s ability to mimic malignancy can be very problematic in a clinical setting. A multidisciplinary approach and an algorithmic diagnostic evaluation are the most befitting clinical strategy for bladder endocervicosis. This includes a combination of medical imaging, urological intervention, and histopathological evaluation. In this case, MRI scan of the pelvis revealed a thickened bladder wall and scar tissue within the vesicouterine space connecting the lower uterine segment to the posterior dome of the urinary bladder with no discrete intraluminal mass visualized. A more recent case of BE by Diamantidis et al. showed MRI findings similar to our case [7]. Cystoscopy identified a solid mass on the posterior wall of the bladder with invaginated papillary projections. Additionally, cystoscopy revealed a mural nodule with smooth urothelial mucosa, a finding that also helped differentiate endocervicosis from malignant tumors.
Moreover, gross examination of the bladder endocervicosis presents as a non-encapsulated tumor-like mass or a diffuse area of bladder wall thickening. Lesions typically range in diameter from 1 to 5 cm [3],[8]. They are characteristically mural lesions centered within the muscularis propria and are covered by an unremarkable urothelial mucosa. The cut surface may appear solid and fibrous or may reveal variably sized cystic spaces containing clear, mucinous, or hemorrhagic fluid [3],[8].
The histopathological hallmark feature of endocervicosis is the presence of irregularly distributed endocervical-type glands within the lamina propria (Table 2). The absence of nuclear atypia, pleomorphism, and mitotic activity rules out malignancy [1],[2],[9]. The architectural pattern may suggest invasive carcinoma, but unlike adenocarcinoma, endocervicosis does not provoke a desmoplastic stromal response. In chronic cases, reported literature documented incidences of concurrent peri-glandular fibrosis and edema. Ruptured glands may lead to extravasated mucin.
Immunohistochemistry is critical in the diagnostic algorithm of endocervicosis and differentiating it from malignancy mimickers. Estrogen receptors (ERs), progesterone receptors (PRs), and PAX8 raise suspicion that the lesion is of Müllerian origin. Cytokeratin 7, Cytokeratin 20, and GATA3 are critical in differentiating endocervicosis from urothelial carcinoma with glandular differentiation (Table 3). The term Müllerianosis is used when there is a combination of several different types of Müllerian epithelium [10]. CDX2−/ER+ is the usual immunohistochemical profile of endocervicosis as seen in our case, which is helpful in differentiating it from intestinal metaplasia CDX2+/ER− in small bladder biopsies [10].
Surgical intervention, such as transurethral resection of bladder tumor (TURBT), is effective for smaller lesions. At the same time, partial cystectomy is reserved for larger and anatomically complex lesions [9]. Transurethral resection of bladder tumor provides a dual diagnostic and interventional utility to endocervicosis. Lesions that are easily accessible through the urethra largely benefit from this dual element [9].
Bladder endocervicosis recurrence is generally rare, but it can occur due to an underlying hormonal link, such as during pregnancy, following transurethral resection [11]. Clinically, the literature reports no standardized follow-up regimen. However, imaging and endoscopic surveillance are recommended due to bladder endocervicosis’s potential for recurrence and its tendency to mimic adenocarcinoma [2].
Conclusion
This case of bladder endocervicosis highlights the importance of a high index of suspicion in the differential diagnosis work-up for chronic pelvic pain in women with a history of pelvic surgery or cesarean sections. Albeit a benign condition, it can cause vexatious symptoms such as long-standing pelvic pain with voiding. An effective step-wise multidisciplinary approach will help distinguish bladder endocervicosis from its differential counterparts, including most notably the malignancy mimickers. As of now, the prognosis is excellent. However, future research should elucidate the outcomes of long-term follow-up studies further to investigate the potential transformation of the lesion into malignancy. Moreover, a more in-depth analysis of the specific immunopathological mechanisms on a molecular level with the metaplastic pathogenesis theory should be explored. Dissemination of clinical awareness on how to maneuver this challenging case will mitigate misdiagnosis of malignancy and prevent unnecessary aggressive surgical interventions.
REFERENCES
1.
Clement PB, Young RH. Endocervicosis of the urinary bladder. A report of six cases of a benign Müllerian lesion that may mimic adenocarcinoma. Am J Surg Pathol 1992;16(6):533–42. [CrossRef]
[Pubmed]

2.
Santos-Lopes S, Cebola A, Afonso A, Ferrito F. Bladder endocervicosis – A rare diagnosis with a silent presentation. Urol Ann 2017;9(4):400–2. [CrossRef]
[Pubmed]

3.
Raspollini MR, Lopez-Beltran A. Endocervicosis. In: Raspollini MR, Lopez-Beltran A, editors. Uropathology. Encyclopedia of pathology. Cham: Springer; 2020. [CrossRef]

4.
Olivia Vella JE, Nair N, Ferryman SR, Athavale R, Latthe P, Hirschowitz L. Müllerianosis of the urinary bladder. Int J Surg Pathol 2011;19(4):548–51. [CrossRef]
[Pubmed]

5.
Cheah PL, Looi LM, Lee GEG, Teoh KH, Mun KS, Nazarina AR. Unusual finding of endocervical-like mucinous epithelium in continuity with urothelium in endocervicosis of the urinary bladder. Diagn Pathol 2011;6:56. [CrossRef]
[Pubmed]

7.
Diamantidis D, Tsakaldimis G, Georgellis C, Lailisidis S, Panagiotopoulos N, Kafalis C, et al. Clinical aspects and therapeutic strategy in a case of urinary bladder endocervicosis. Prz Menopauzalny 2024;23(2):109–12. [CrossRef]
[Pubmed]

8.
Eskridge MR, Rovner ES, Payne KD, Workman RB, Curry NS. MRI of endocervicosis: An unusual cause of clustered periurethral cystic masses involving the bladder. AJR Am J Roentgenol 2007;188(2):W147–9. [CrossRef]
[Pubmed]

9.
Heretis J, Stamatiou K, Papadimitriou V, Giannikaki E, Stathopoulos E, Sofras F. Endocervicosis of the bladder: Report of a case and review of the current literature. Int J Clin Exp Pathol 2009;2(1):91–4.
[Pubmed]

10.
11.
Whiting D, Ali A. Bladder endocervicosis: Recurrent presentations during pregnancy. Urol Ann 2021;13(3):323–5. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Acknowledgments
Medical imaging acknowledgement. Dr. Oleg Mironov, MD, FRCPC, Department of Medical Imaging, McMaster University, Hamilton, Ontario.
Author ContributionsMoaz Alowami - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Oluwaseun Adelekan - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Lorraine Min-Shan Liang - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Pooja Vasudev - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionDr. Pooja Vasudev is the guarantor of submission and accepts full responsibility for the integrity of the work, had access to all the data in the study, and controlled the decision to publish.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2026 Moaz Alowami et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.