|
Case Report
Aspergilloma mimicking postoperative recurrence of squamous cell lung cancer: A case report
1 Faculty of Medicine, University of Belgrade, 11000 Belgrade, Serbia
2 Clinic for Thoracic Surgery, University Clinical Center of Serbia, 11000 Belgrade, Serbia
3 Clinic for Thoracic Surgery, University Clinical Center Niš, 18000 Niš, Serbia
4 Center for Radiology, University Clinical Center of Serbia, 11000 Belgrade, Serbia
5 Clinic for Pulmonology, University Clinical Center of Serbia, 11000 Belgrade, Serbia
Address correspondence to:
Stefan Stevanović
Clinic for Thoracic Surgery, University Clinical Center Niš, 10 Vojislava Ilića Street, 18000 Niš,
Serbia
Message to Corresponding Author
Article ID: 101516Z01MS2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Savić M, Stevanović S, Garabinović Ž, Čolić N, Lukić K, Marić D. Aspergilloma mimicking postoperative recurrence of squamous cell lung cancer: A case report. Int J Case Rep Images 2025;16(2):20–24.ABSTRACT
Introduction: Pulmonary aspergilloma (PA) is a common clinical manifestation of lung infection caused by Aspergillus species. In addition to tuberculosis and chronic obstructive pulmonary disease, which are considered the most frequent underlying conditions, lung cancer is also recognized as an important etiopathogenetic factor.
Case Report: A chest computed tomography (CT) scan performed six months after thoracoscopic right upper lobectomy for primary squamous cell lung carcinoma (three months after completing chemotherapy) revealed a spiculated lesion in the apical segment of the ipsilateral lower pulmonary lobe, measuring 28×39×42 mm. Fine-needle aspiration biopsy (FNAB) and serological testing confirmed Aspergillus species as the etiological agent of the pulmonary mass. The patient underwent conservative antifungal therapy, starting with 14 days of intravenous voriconazole during hospitalization, followed by oral itraconazole for six weeks post-discharge. Notably, he remained asymptomatic both prior to diagnosis and throughout treatment. Follow-up chest CT scans revealed complete resolution of the pulmonary aspergilloma, with residual calcified fibrous tissue consistent with chronic post-treatment changes.
Conclusion: This case underscores the importance of considering PA in the differential diagnosis of new lung lesions detected after lung cancer surgery, given its ability to closely mimic tumor recurrence.
Keywords: Antifungal therapy, Chronic pulmonary aspergillosis, Lung cancer, Radiological presentation
Introduction
Pulmonary aspergilloma (PA) is one of the five acknowledged clinicopathological forms of chronic pulmonary aspergillosis (CPA) [1],[2] and a frequent clinical presentation of lung infection caused by Aspergillus species. Its defining feature is the presence of a viable fungal mass inside a previously existing lung cavity. Usually, this mass is made up of fungal colonies, most notably A. fumigatus (in about 95% of cases), as well as inflammatory cells, fibrin, blood, and mucus [2],[3].
Pulmonary aspergilloma is slightly more common in males, with a median age of 65 years at diagnosis. Most patients lived either in farming areas or in large cities [4]. This condition is commonly associated with pre-existing pulmonary comorbidities, such as tuberculosis (reported in 25–80% of cases), chronic obstructive pulmonary disease (COPD), non-tuberculous mycobacterial infections (NTMB), bronchiectasis, sarcoidosis, lung abscesses, and necrotic lung cancer [3],[4],[5].
This report presents the case of a patient diagnosed with PA six months after surgical treatment for primary squamous cell lung carcinoma (SQCLC), although the radiographic appearance of a mass observed in the right lower lung lobe was most indicative of tumor relapse.
Case Report
A 69-year-old patient, a former smoker, comes for a surgical examination, in order to assess the possibility of surgical treatment of histopathologically confirmed primary SQCLC. The patient reported a 5-month history of productive cough. His medical history included arterial hypertension, type 2 diabetes mellitus, and benign prostatic hyperplasia. He claimed that he had never experienced tuberculosis or any other respiratory comorbidities. Transbronchial biopsy tissue samples were histopathologically analyzed to determine the diagnosis of SQCLC. The patient had not undergone any neoadjuvant treatment. The chest X-ray (Figure 1) shows a soft tissue mass with an oval shape measuring up to 40 mm in diameter in the center right pulmonary field, next to the lateral chest wall. Contrast-enhanced CT of the chest (CECT) more precisely identified an excavated tumor with thickened walls and lobulated contours, situated in the upper lobe of the right lung, measuring 41×37×30 mm (Figure 2A and Figure 2B).
Following a thorough preoperative assessment and after obtaining informed consent from the patient, surgical intervention was carried out. Two months after the diagnosis, the patient had a right upper lobectomy using a uniportal thoracoscopic technique. On the fourth day following surgery, the patient was released after a smooth perioperative and postoperative course. According to the multidisciplinary tumor board’s advice, postoperative management included 3 cycles of adjuvant chemotherapy using cisplatin and etoposide. The resected lung tissue’s histopathological analysis revealed a primary SQCLC that was invasive and moderately differentiated. All dissected lymph nodes (interlobar, hilar, subcarinal, and lower paratracheal) had no metastases, and the surgical borders were clear (R0 resection). The tumor was classified as IB (pT2aN0Mx) in accordance with the 9th edition of the TNM classification for lung cancer [6].
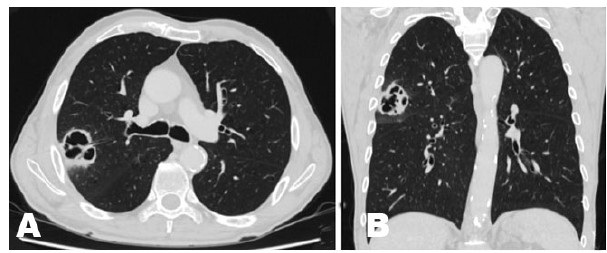
Six months after surgery (three months after chemotherapy), a follow-up chest CECT scan revealed a recently formed oval lesion with spiculated borders in the superior segment (S6) of the ipsilateral right lower lobe, measuring 28×39×42 mm, as well as two neighboring nodules near the interlobar area (Figure 3A). The patient was then admitted to the Pulmonology Clinic for additional testing and therapy.
Fine-needle aspiration biopsy (FNAB) of the lesion excluded tumor recurrence, which had been the initial suspicion due to the lesion‘s radiographic features, and confirmed the presence of fungal elements in the lung tissue, consisting of granulation tissue and fungal colonies. Serologic testing supported the diagnosis of an Aspergillus infection (anti-Aspergillus IgM: 836 U/mL; IgG negative).
The patient was treated conservatively: initially with intravenous voriconazole (200 mg twice on the first day, followed by 200 mg once daily for 13 days), and continued with oral itraconazole (200 mg daily) for six weeks following discharge. Throughout hospitalization and treatment, the patient remained asymptomatic. Hematological parameters, including neutrophil and eosinophil counts, as well as standard biochemical markers such as C-reactive protein, plasma procalcitonin, IL-6, and erythrocyte sedimentation rate, remained within normal reference ranges.
Over a 15-month follow-up period, control CECT scans demonstrated complete resolution of the PA, with residual calcified fibrous tissue replacing the previously observed lesion (Figure 3B, Figure 3C, Figure 3D, Figure 3E, Figure 3F). Also, there were no signs of SQCLC recurrence.
Discussion
Although tuberculosis and COPD are among the most common pulmonary conditions associated with the development of PA, recent studies have also identified lung cancer as an important etiopathogenetic factor. Several mechanisms have been proposed to explain the potential coexistence of these two diseases: (I) structural damage to the lung parenchyma caused by the malignant process (e.g., inflammation, ischemic necrosis); (II) surgical intervention itself (e.g., dead space, iatrogenic lung injury, prolonged air leak, bronchopleural fistula); and (III) post-radiation cavitation, among others [5],[7],[8],[9].
In patients with lung cancer, Shin et al. [7] found a 10-year incidence of PA of 3.5%. It is interesting to note that, in line with the current case, they discovered that patients who had surgery for SQCLC had a considerably higher incidence of PA (41.1% of cases) than those with other histological subtypes of lung cancer [7]. The histopathological features of SQCLC, such as its rapid growth rate, central ischemic necrosis, and resulting development of cavitary lesions, may account for this link [7],[10]. The development of PA may result from the secondary colonization of these cavitations by fungi.
Additional risk factors for PA in patients with lung cancer include male sex, smoking history, lower body mass index, immunosuppression, COPD, interstitial lung disease, thoracotomy (which carries a ninefold higher risk compared to thoracoscopic approaches), late postoperative complications (such as pneumonia or atelectasis), and concomitant chemoradiotherapy [5],[7],[8],[9].
The most common clinical manifestations of PA are fever, malaise, weight loss, productive cough, and hemoptysis, which can occasionally be massive and fatal [11]. However, PA can be present as an indolent form of aspergillosis, particularly in immunocompetent, asymptomatic patients, as seen in this case.
A combination of clinical presentation, radiological results, and laboratory tests, such as fungal cultures, serological testing, and histopathological analysis, is used to diagnose PA [11],[12]. The identification of anti-Aspergillus IgG antibodies in serum is indicative of PA, but their absence does not rule it out. According to a study by Zhu et al. [12], just 41.7% of PA patients had measurable anti-Aspergillus IgG antibodies.
Management of PA can be either surgical or conservative. In 7–10% of cases, spontaneous resolution has been documented [3]. Posterolateral thoracotomy with extrapleural dissection remains the standard open surgical approach, offering direct visualization of the lesion, controlled pleuro-pulmonary dissection, effective lung palpation, and prompt hemostasis [13],[14]. Lobectomy is generally considered the preferred extent of resection [7],[8],[15], although segmentectomy or wedge resection may be sufficient in asymptomatic patients with small (<3 cm), peripheral lesions [2],[4]. In our case, surgical intervention was not performed. Following consultation with a thoracic surgeon, antifungal therapy was initiated with voriconazole, followed by itraconazole. The patient responded well to the treatment, with complete radiological resolution of the lesion.
In terms of pharmacological therapy, the Infectious Diseases Society of America (IDSA) guidelines emphasize the usage of itraconazole and voriconazole as first-line drugs for PA [16]. Studies assessing the effectiveness of these two agents in CPA show that itraconazole has a clinical response rate of around 76.5% and voriconazole has a clinical response rate of around 61% [17],[18].
The prognosis of lung cancer in the context of PA is contingent upon a number of variables. According to Jiang et al. [1], older age, male sex, diabetes mellitus, and sublobar resection were all significantly linked to worse outcomes. Regarding PA, the retrospective study conducted by Bo-Guen Kim et al. [19] indicates that the development of CPA did not affect lung cancer mortality in the multivariable analysis.
Conclusion
Pulmonary aspergilloma and SQCLC share several risk factors, as well as overlapping clinical and radiological features. Given that PA can closely mimic primary SQCLC or its recurrence, both clinically and radiographically, this case underscores the importance of including PA in the differential diagnosis of new or recurrent pulmonary lesions—particularly in patients with aforementioned risk factors.
This case report may be of particular interest to infectious disease specialists, pulmonologists, thoracic surgeons, as well as general practitioners, medical residents, and students.
REFERENCES
1.
Jiang C, Dai J, Bao Y, Jiang G, Zhu Y, Zhang P. Surgical treatment of pulmonary aspergilloma: A 13-year experience from a single clinical center. Ann Thorac Surg 2022;114(1):311–8. [CrossRef]
[Pubmed]

2.
Yuan P, Cao JL, Huang S, Zhang C, Bao FC, Hu YJ, et al. Sublobar resection for pulmonary aspergilloma: A safe alternative to lobectomy. Ann Thorac Surg 2017;103(6):1788–94. [CrossRef]
[Pubmed]

3.
Lang M, Lang AL, Chauhan N, Gill A. Non-surgical treatment options for pulmonary aspergilloma. Respir Med 2020;164:105903. [CrossRef]
[Pubmed]

4.
Tashiro M, Takazono T, Izumikawa K. Chronic pulmonary aspergillosis: Comprehensive insights into epidemiology, treatment, and unresolved challenges. Ther Adv Infect Dis 2024;11:20499361241253751. [CrossRef]
[Pubmed]

5.
Harmouchi H, Sani R, Issoufou I, Lakranbi M, Ouadnouni Y, Smahi M. Pulmonary aspergilloma: From classification to management. Asian Cardiovasc Thorac Ann 2020;28(1):33–8. [CrossRef]
[Pubmed]

6.
Son JW, Lee J, Jeon JH, Cho S, Jung W, Shih BCH, et al. Validation of IASLC 9th edition TNM classification for lung cancer: Focus on N descriptor. BMC Cancer 2024;24(1):1460. [CrossRef]
[Pubmed]

7.
Shin SH, Kim BG, Kang J, Um SW, Kim H, Kim HK, et al. Incidence and risk factors of chronic pulmonary aspergillosis development during long-term follow-up after lung cancer surgery. J Fungi (Basel) 2020;6(4):271. [CrossRef]
[Pubmed]

8.
Tamura A, Suzuki J, Fukami T, Matsui H, Akagawa S, Ohta K, et al. Chronic pulmonary aspergillosis as a sequel to lobectomy for lung cancer. Interact Cardiovasc Thorac Surg 2015;21(5):650–6. [CrossRef]
[Pubmed]

9.
Wu T, Li P, Wang M, Wang Q, Shi Y, Su X. Pulmonary solid tumor with coexisting pulmonary aspergillosis: Case reports and literature review. Clin Respir J 2017;11(1):3–12. [CrossRef]
[Pubmed]

10.
Parkar AP, Kandiah P. Differential diagnosis of cavitary lung lesions. J Belg Soc Radiol 2016;100(1):100. [CrossRef]
[Pubmed]

11.
Barac A, Vujovic A, Drazic A, Stevanovic G, Paglietti B, Lukic K, et al. Diagnosis of chronic pulmonary aspergillosis: Clinical, radiological or laboratory? J Fungi (Basel) 2023;9(11):1084. [CrossRef]
[Pubmed]

12.
Zhu RS, Zhou LH, Cheng JH, Luo Y, Qiu WJ, Huang JT, et al. Diagnostic laboratory features and performance of an Aspergillus IgG lateral flow assay in a chronic pulmonary aspergillosis cohort. Microbiol Spectr 2023;11(3):e0026423. [CrossRef]
[Pubmed]

13.
Chen QK, Jiang GN, Ding JA. Surgical treatment for pulmonary aspergilloma: A 35-year experience in the Chinese population. Interact Cardiovasc Thorac Surg 2012;15(1):77–80. [CrossRef]
[Pubmed]

14.
Harmouchi H, Lakranbi M, Issoufou I, Ouadnouni Y, Smahi M. Pulmonary aspergilloma: Surgical outcome of 79 patients in a Moroccan center. Asian Cardiovasc Thorac Ann 2019;27(6):476–80. [CrossRef]
[Pubmed]

15.
Balasubbiah N, Saeteng S, Siwachat S, Thuropathum P, Tantraworasin A. Outcomes of pulmonary resection in pulmonary aspergilloma: A retrospective cohort study in a single tertiary-care hospital in Northern Thailand. Asian J Surg 2024;47(7):3033–8. [CrossRef]
[Pubmed]

16.
Patterson TF, Thompson GR 3rd, Denning DW, Fishman JA, Hadley S, Herbrecht R, et al. Practice guidelines for the diagnosis and management of aspergillosis: 2016 update by the Infectious Diseases Society of America. Clin Infect Dis 2016;63(4):e1–60. [CrossRef]
[Pubmed]

17.
Agarwal R, Vishwanath G, Aggarwal AN, Garg M, Gupta D, Chakrabarti A. Itraconazole in chronic cavitary pulmonary aspergillosis: A randomised controlled trial and systematic review of literature. Mycoses 2013;56(5):559–70. [CrossRef]
[Pubmed]

18.
Jain LR, Denning DW. The efficacy and tolerability of voriconazole in the treatment of chronic cavitary pulmonary aspergillosis. J Infect 2006;52(5):e133–7. [CrossRef]
[Pubmed]

19.
Kim BG, Choi YS, Shin SH, Lee K, Um SW, Kim H, et al. Mortality and lung function decline in patients who develop chronic pulmonary aspergillosis after lung cancer surgery. BMC Pulm Med 2022;22(1):436. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Milan Savić - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Stefan Stevanović - Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Željko Garabinović - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Nikola Čolić - Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Katarina Lukić - Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Dragana Marić - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Milan Savić et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.