|
Case Report
Spinal meningeal melanocytoma with direct bone metastasis: A case report and literature review
1 Department of Neurosurgery, Aichi Medical University, Nagakute-shi, Aichi-ken, Japan
Address correspondence to:
Toshiaki Inomo
#202 Anju6 2-502-2 Sakuta, Nagakute-shi, Aichi-ken 480-1153,
Japan
Message to Corresponding Author
Article ID: 101415Z01TI2023
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Inomo T, Aoyama M, Maejima R, Hara M. Spinal meningeal melanocytoma with direct bone metastasis: A case report and literature review. Int J Case Rep Images 2023;14(2):80–84.ABSTRACT
Meningeal melanocytoma is a rare benign tumor of the central nervous system. An intradural extramedullary tumor at C2 was accidentally discovered in a 58-year-old woman six years ago. The lesion was well-circumscribed mass on computed tomography and magnetic resonance imaging. We predicted the tumor to be a meningioma. We performed tumor resection because its gradual growth compressed the spinal cord. Intraoperatively, the tumor was dark brown and the nearby dura matter and the lamina had a pigmented lesion suggesting direct invasion. After gross total removal, the tumor was diagnosed as meningeal melanocytoma by pathological examination. Meningeal melanocytoma with direct bone metastasis are rare. We present this case with reference to previous literature.
Keywords: Direct bone metastasis, Melanoma, Radiological features, Spinal meningeal melanocytoma, Spinal tumor
Introduction
Melanocytomas comprise <0.1% of all tumors in the central nervous system (CNS) [1]; meningeal melanocytoma is a rare benign tumor derived from leptomeningeal melanocytes [2]. Almost all of these tumors are found at the posterior fossa or upper spinal level [3],[4]. The most common image appearance is as a well-defined intradural extramedullary mass; other differential diagnoses are meningioma or schwannoma [3],[5]. Typical magnetic resonance imaging (MRI) findings are hyperintense on T1-weighted images (T1WI) and hypointense on T2-weighted images (T2WI), with unclear contrast enhancement [3]. In pathology, melanoma should be excluded. Relatively good prognosis can be expected after total resection. However, some cases report an aggressive course of rapid growth, recurrence, and transformation to malignant melanoma [6],[7]. Accordingly, careful observation is essential. We report a rare case of meningeal melanocytoma with direct bone invasion at the C2 level that was completely removed by surgery.
Case Report
A 52-year-old woman with no previous medical history was accidentally diagnosed with a cervical spinal tumor at C2 level when injured in a traffic accident (Figure 1A). Since then, she had been complaining of numbness of the left limbs but there was no clear association with the lesion. The tumor kept gradually growing and the spinal cord became compressed over a 6-year follow-up, without symptom exacerbation. She planned to undergo tumor removal in August 2022. Preoperatively, the mass was well-circumscribed, and no calcification was observed by computed tomography (CT). A cervical spinal CT revealed that it was intradural extramedullary, hyperintense on T1WI, hypointense on T2WI, with unclear contrast enhancement (Figure 1B, Figure 1C, Figure 1D, Figure 1E, Figure 1F, Figure 1G, Figure 1H). There was no lesion on head MRI.
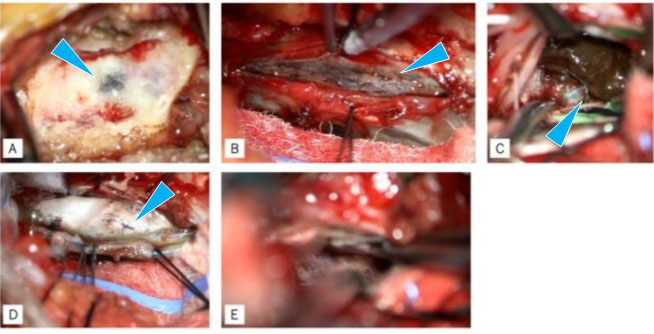
A posterolateral approach was performed to resect the tumor. We approached the dura matter by C2 partial laminectomy and performed an incision. Intraoperatively, we found that the tumor was dark brown, and the lamina of the C2, dura, and pia nearby were partially pigmented. We removed the tumor piece by piece due to its softness and adhesion to dura, until achieving gross total resection (Figure 2A, Figure 2B, Figure 2C, Figure 2D, Figure 2E). After resection, the dural attachment was coagulated and the dura was sutured water-tight.
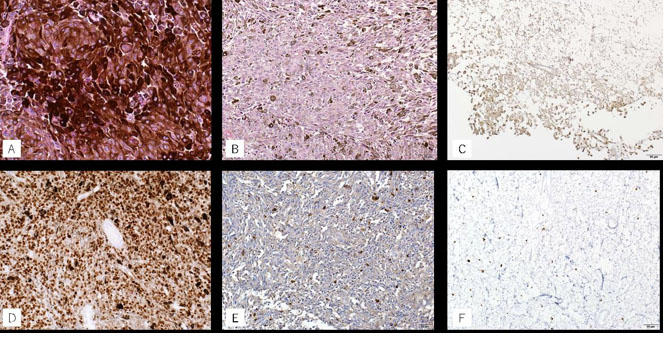
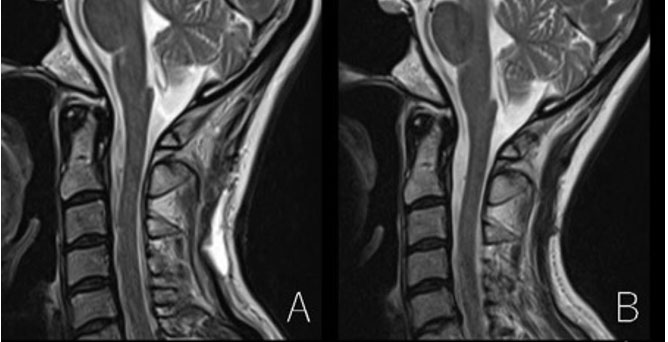
In pathological examination, proliferation of tumor cells with high melanin deposition was found in the tumor and in the pigmented lesion of the vertebral arch and dura (Figure 3A). There was slight cellular atypism but no atypical mitotic figure (0/10 high power field) but no clear necrosis. Immunohistochemistry analysis showed positive results for SRY-related high-mobility-group/HMG box 10 (SOX-10), S-100 protein, and human melanoma black-45 (HMB-45) (Figure 3B, Figure 3C, Figure 3D, Figure 3E). The Ki-67 index was <2% (Figure 3F). The tumor was diagnosed as meningeal melanocytoma as there were insufficient findings to consider it as malignant melanoma. The patient is followed up regularly but not receiving radiation or chemotherapy. An MRI at three months after surgery showed no evidence of tumor recurrence (Figure 4A and Figure 4B).
Discussion
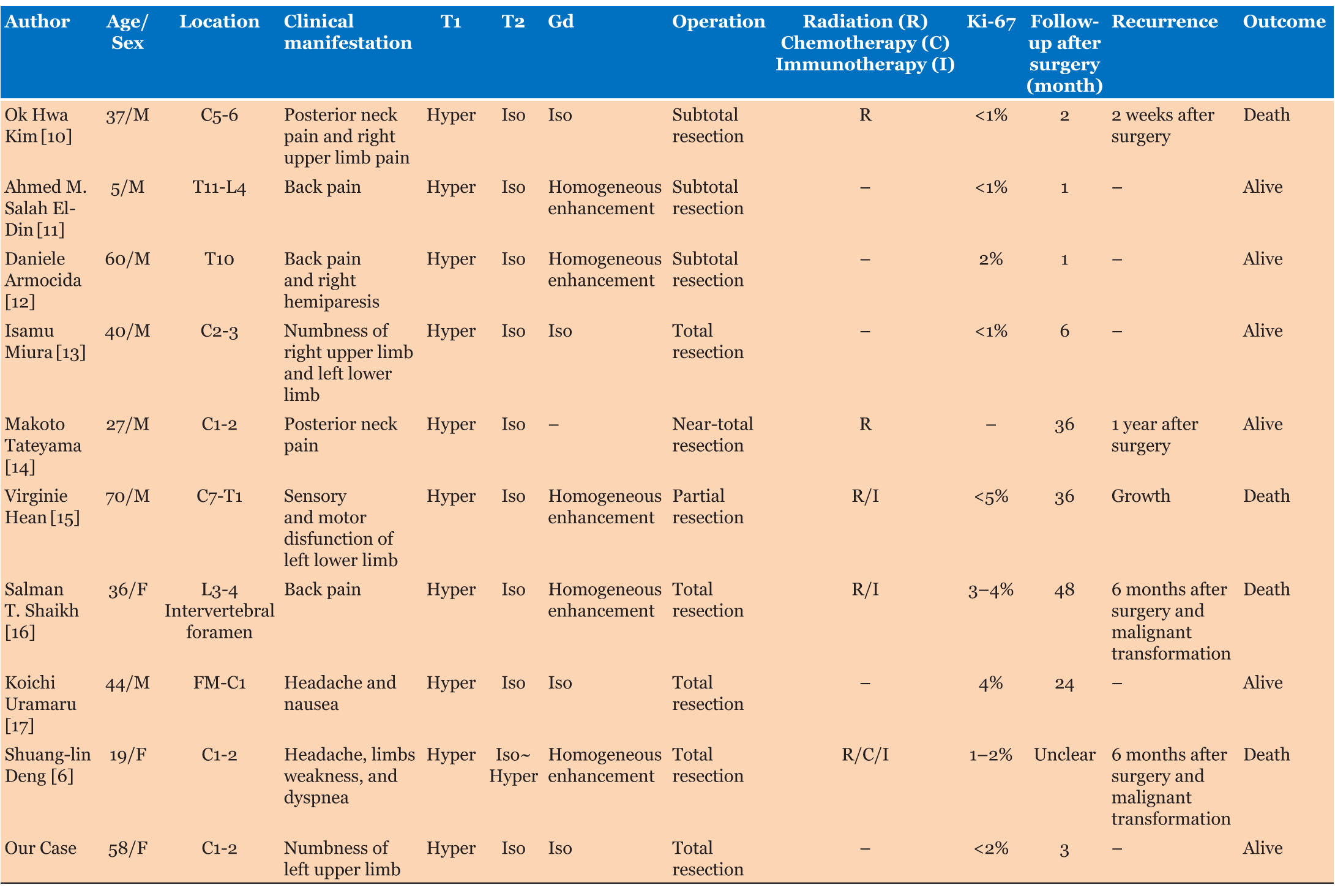
In 1972, Limas and Tio first reported a melanocytic tumor in the craniocervical junction [8]. According to the 2016 World Health Organization classification of CNS tumors, primary melanocytic tumors of the CNS are classified as: (1) meningeal melanocytosis (Grade 0), (2) meningeal melanocytoma (Grade 1), (3) meningeal melanoma (Grade 3), and (4) meningeal melanomatosis (Grade 3). Surgical removal is recommended as primary treatment [9]. Gross total removal is not difficult for intradural extramedullary tumors. The radiographic features and treatment of primary CNS melanocytoma from 10 case reports are summarized in Table 1. Most cases (9/10) achieved gross total resection or subtotal/near-total resection. In one case of partial resection, the patient was treated by radiation, immunotherapy, and second surgery, but he finally expired three years after diagnosis [15]. Two cases suffered recurrence and malignant transformation despite gross total resection and underwent chemotherapy. However, they showed resistance to the treatment [6],[16]. To the best of our knowledge, there are few reports of a good course with radiation and chemotherapy. We highly recommend total resection as first treatment of meningeal melanocytoma before tumor growth to prevent complications. Radiation and chemotherapy should be recognized as adjunctive treatment when total resection is not achieved or in case of tumor recurrence. Recently, immunotherapy with ipilimumab (anti-cytotoxic T1-Lymphocyte-assosiated protein drug) or nivolumab (anti-programmed cell death protein-1 drug) showed good prognosis in melanoma [18]. The suggested effectiveness of those treatments for meningeal melanocytoma or melanoma warrants further research [16],[18]. Histopathologic and immunohistochemical examination is essential to diagnose meningeal melanocytoma. Immunohistochemical staining indicates that melanocytoma is positive for S-100 and HMB-45 but negative for epithelial membrane antigen [3],[19],[20]. In addition, the Ki-67/MIB-1 index is generally low (<5%), in contrast with the high score found in melanoma [2],[3].
In our case, the tumor was well-circumscribed, hyperintense, on T1WI and hypointense on T2WI, with unclear contrast enhancement. Its slow growth as well as the histopathologic and immunohistochemical findings of low Ki-67 index and no abnormal mitosis/necrosis suggested meningeal melanocytoma. In contrast, the intraoperative findings of direct bone metastasis cannot exclude melanoma. Previous research indicated a transformation of melanocytoma into melanoma [6],[7],[16], so we should consider radiotherapy depending on the disease course. Despite some reports of dural invasion in meningeal melanocytoma, we found no report of direct bone invasion; thus, the present case is rare.
Conclusion
We report a rare case of meningeal melanocytoma in the upper cervical spine. Before surgery, we considered the tumor as a meningioma. It was difficult to identify as meningeal melanocytoma from images and clinical symptoms, as intraoperative findings, pathological examination, and immunostaining are essential to diagnose it. We recommend complete excision at an early stage as first choice treatment to avoid complications and transformation to malignant melanoma. In the present case, the gradual growth and pathological appearance indicates benign and good prognosis from surgical total resection. However, the direct bone invasion makes local recurrence possible, so regular follow-up is mandatory.
REFERENCES
1.
Liubinas SV, Maartens N, Drummond KJ. Primary melanocytic neoplasms of the central nervous system. J Clin Neurosci 2010;17(10):1227–32. [CrossRef]
[Pubmed]

2.
Brat DJ, Giannini C, Scheithauer BW, Burger PC. Primary melanocytic neoplasms of the central nervous systems. Am J Surg Pathol 1999;23(7):745–54. [CrossRef]
[Pubmed]

3.
Yang C, Fang J, Li G, et al. Spinal meningeal melanocytomas: Clinical manifestations, radiological and pathological characteristics, and surgical outcomes. J Neurooncol 2016;127(2): 279–86. [CrossRef]
[Pubmed]

4.
Chen CJ, Hsu YI, Ho YS, Hsu YH, Wang LJ, Wong YC. Intracranial meningeal melanocytoma: CT and MRI. Neuroradiology 1997;39(11):811–4. [CrossRef]
[Pubmed]

5.
Ahluwalia S, Ashkan K, Casey ATH. Meningeal melanocytoma: Clinical features and review of the literature. Br J Neurosurg 2003;17(4):347–51. [CrossRef]
[Pubmed]

6.
Deng SL, Wang YB, Wang DH, Zhan S, Jing Y, Guan Y. Malignant transformation and metastatic spread of dumbbell-shaped meningeal melanocytoma of the cervical spine: A case report and literature review. Front Surg 2022;9:789256. [CrossRef]
[Pubmed]

7.
Perrini P, Caniglia M, Pieroni M, Castagna M, Parenti GF. Malignant transformation of intramedullary melanocytoma: Case report. Neurosurgery 2010;67(3):E867–9. [CrossRef]
[Pubmed]

8.
Limas C, Tio FO. Meningeal melanocytoma (“melanotic meningioma”). Its melanocytic origin as revealed by electron microscopy. Cancer 1972;30(5):1286–94. [CrossRef]
[Pubmed]

9.
Rades D, Schild SE, Tatagiba M, Molina HA, Alberti W. Therapy of meningeal melanocytomas. Cancer 2004;100(11):2442–7. [CrossRef]
[Pubmed]

10.
Kim OH, Kim SJ, Choo HJ, Lee SJ, Lee IS, Kim JY, Kim H. Spinal meningeal melanocytoma with benign histology showing leptomeningeal spread: Case report. Korean J Radiol 2013;14(3):470–6. [CrossRef]
[Pubmed]

11.
Salah El-Din AM, Aboul-Ela HM, Alsawy MF, Koheil A, Ashry AH. Spinal meningeal melanocytoma in a 5-year-old child: A case report and review of literature. Egypt J Neurol Psychiatr Neurosurg 2018;54(1):13. [CrossRef]
[Pubmed]

12.
Armocida D, Pesce A, Berra LV, Marzetti F, Antonelli M, Santoro A. Intradural extramidullary dorsal melanocytoma in the adult: Case report and review of the literature. J Clin Neurosci 2019;62:248–53. [CrossRef]
[Pubmed]

13.
Miura I, Kubota M, Momosaki O, Nyui M, Takebayashi K, Kawamata T, Yuzurihara M. A rapidly growing cervical meningeal melanocytoma with a dumbbell-shaped extension. World Neurosurg 2020;134:90–3. [CrossRef]
[Pubmed]

14.
Tateyama M, Fujimoto T, Nakamura T, Miyamoto T. Meningeal melanocytoma occurring at epidural region of the cervical spine. Spine Surg Relat Res 2020;4(4):377–9. [CrossRef]
[Pubmed]

15.
Hean V, Bouleftour W, Ramirez C, Forest F, Boutet C, Rivoirard R. Nivolumab as adjuvant treatment for a spinal melanocytoma: A case report. Medicine (Baltimore) 2021;100(19):e25862. [CrossRef]
[Pubmed]

16.
Shaikh ST, Gupta GS, Mohanty CB, Deopujari CE. Spinal nerve root extradural melanocytoma progressing to malignant melanoma: A case report with review of literature. Asian J Neurosurg 2021;16(2):394–7. [CrossRef]
[Pubmed]

17.
Uramaru K, Sakata K, Shimohigoshi W, Kawasaki T, Manaka H. Primary meningeal melanocytoma located in the craniovertebral junction: A case report and literature review. NMC Case Rep J 2021;8(1):349–54. [CrossRef]
[Pubmed]

18.
Wolchok JD, Chiarion-Sileni V, Gonzalez R, et al. Overall survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med 2017;377(14):1345–56. [CrossRef]
[Pubmed]

19.
Lee JK, Rho YJ, Jeong DM, Rhim SC, Kim SJ. Diagnostic clue of meningeal melanocytoma: Case report and review of literature. Yonsei Med J 2017;58(2):467–70. [CrossRef]
[Pubmed]

20.
Clarke DB, Leblanc R, Bertrand G, Quartey GR, Snipes GJ. Meningeal melanocytoma. Report of a case and a historical comparison. J Neurosurg 1998;88(1):116–21. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Toshiaki Inomo - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Masahiro Aoyama - Conception of the work, Design of the work, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ryuya Maejima - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Masahito Hara - Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2023 Toshiaki Inomo et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.