|
Case Report
A rapidly fatal case of black esophagus in a patient with multiple co-morbidities
1 Resident, Department of Internal Medicine, West Virginia University School of Medicine, Morgantown, WV, USA
2 Resident, Department of Internal Medicine, Saint John’s Episcopal Hospital, Far Rockaway, NY, USA
3 Resident, Department of Pathology, West Virginia University School of Medicine, Morgantown, WV, USA
4 Gastroenterologist, Department of Gastroenterology, University of Tripoli, Tripoli, Libya
Address correspondence to:
Olanrewaju Adeniran
1 Medical Center Drive, Morgantown, WV 26506,
USA
Message to Corresponding Author
Article ID: 101495Z01OA2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Adeniran O, Marwizi F, Kirkpatrick J, Adekolu A, Sulaiman A, Cohen EM, Zitun M. A rapidly fatal case of black esophagus in a patient with multiple co-morbidities. Int J Case Rep Images 2025;16(1):33–37.ABSTRACT
Introduction: Black esophagus, or acute esophageal necrosis (AEN), is a rare and severe condition characterized by diffuse black discoloration of the esophageal mucosa. It predominantly affects males in their seventh decade and is characterized by diffuse circumferential black discoloration of the esophageal mucosa. The literature describes the disease entity as multifactorial in origin, although tissue hypoperfusion is predominantly a common association. A high index of suspicion and timely identification of the disease combined with appropriate interventions, especially in elderly patients with multiple risk factors, is necessary to reduce the rate of adverse outcomes and high mortality rates associated with AEN.
Case Report: A 73-year-old male with poorly controlled type 2 diabetes mellitus, hypertension, and Barrett’s esophagus presented with massive hematemesis and hemodynamic instability. An urgent endoscopy revealed diffuse black discoloration of the esophageal mucosa with a polypoidal mass at the gastroesophageal junction. The initial biopsy was non-diagnostic, but a repeat biopsy showed poorly differentiated carcinoma. Despite initial recovery and discharge, the patient returned with acute respiratory failure and died within a week.
Conclusion: This case highlights the complex interplay between multiple co-morbidities, including an underlying malignancy and the development of AEN. The case emphasizes the importance of early recognition, prompt intervention, and consistent follow-up care in patients with risk factors for AEN. The challenges in obtaining diagnostic biopsies due to tissue friability suggest potential underdiagnosis of underlying malignancy in AEN cases.
Keywords: Acute esophageal necrosis, Barrett’s esophagus, Black esophagus, Esophageal malignancy, Upper gastrointestinal bleeding
Introduction
Black esophagus, or acute esophageal necrosis (AEN), is a rare and severe condition characterized by diffuse black discoloration of the esophageal mucosa. It has a multifactorial origin, primarily involving tissue hypoperfusion leading to ischemic necrosis of the distal esophagus [1]. According to the literature, the incidence of AEN in patients undergoing esophagogastroduodenoscopy (EGD) ranges between 0.01% and 0.28%. Acute esophageal necrosis predominantly affects men, with a male-to-female ratio of 4:1, and its incidence peaks in the seventh decade, correlating with comorbidities [1]. Since its first description in 1990 by Goldenberg et al., a number of cases have been reported globally [2],[3].
The condition carries a high mortality rate of 30–50%, with survival periods ranging from days to months based on individual patient factors and comorbidities [2],[3],[4]. The pathogenesis involves impaired blood supply, leading to cell death and tissue necrosis, with contributing factors including hypotension, infections, medications, and compromised mucosal integrity. Early recognition and intervention are crucial. Clinical findings can be nonspecific, with most patients presenting with upper gastrointestinal bleeding (UGIB), hematemesis, or melena, and severe cases showing hemodynamic instability and organ dysfunction [1].
This report discusses a rapid fatal case of black esophagus in a patient with poorly controlled diabetes, hypertension, and a history of Barrett’s metaplasia, presenting with massive hematemesis.
Case Report
A 73-year-old man with poorly controlled type 2 diabetes mellitus (T2DM), hypertension (HTN), gastroesophageal reflux disease (GERD) with Barrett’s esophagus, and atrial fibrillation presented to the medical intensive care unit (ICU) from the emergency department (ED) with a 2-hour history of multiple episodes of hematemesis. These began spontaneously while at rest. He had a 3-week history of mild intermittent epigastric pain, nausea, and approximately 15 episodes of clear vomiting, which later progressed to coffee ground emesis. He also reported progressive dysphagia to solids, anorexia, a 40-pound weight loss over two months, palpitations, and melena but no hematochezia.
The patient had a 30-pack-year smoking history but had quit for 33 years. He denied alcohol or substance abuse. His family history was unremarkable. An EGD and biopsy five years prior showed intestinal metaplasia without dysplasia in the distal esophagus, but the patient was lost to follow-up. A barium swallow, a month before presentation, revealed a 9 cm irregular mucosal area in the distal esophagus. Home medications included aspirin, apixaban, metoprolol, pantoprazole, and amlodipine, with questionable compliance
On examination, heart rate was 145 bpm and irregular, respiratory rate was 25 breaths/min, with a blood pressure of 109/40 mmHg. He had mild epigastric tenderness and melena during the digital rectal examination. Laboratory workup revealed hemoglobin of 10.7 g/dL (baseline 13.9 g/dL), white blood cells (WBC) of 18.2 cells/L, lactate of 2.5 mmol/L, and albumin of 2.3 g/dL. The metabolic panel and liver enzymes were unremarkable. Initial management included intravenous fluid resuscitation, antiemetics, analgesics, intravenous (IV) Protonix, a unit of packed red blood cells, IV labetalol, and later an amiodarone drip.
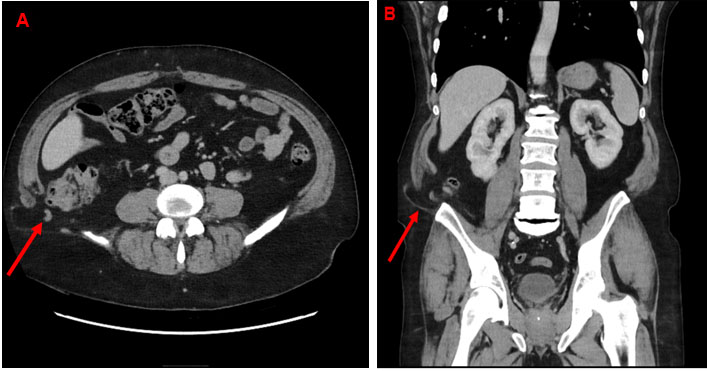
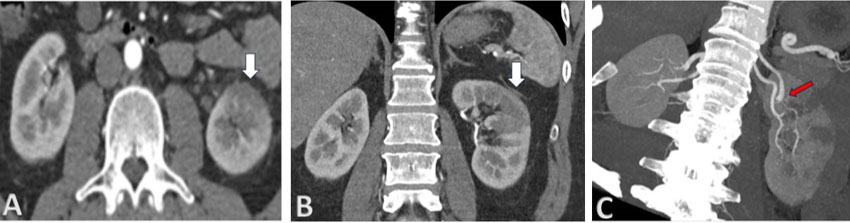
After resuscitation, vitals improved to normal ranges. A computed tomography (CT) scan revealed marked circumferential wall thickening and necrotic intraluminal contents in the mid to lower esophagus—numerous metastatic lesions in the lungs, pleura, mediastinum, and liver (Figure 1). Esophagogastroduodenoscopy showed black, diffusely necrotic mucosa 20 cm from the gastroesophageal junction (GEJ) and a polypoidal mass at the GEJ with normal appearing GEJ and gastric mucosa (Figure 2 and Figure 3). Histopathologic evaluation revealed markedly inflamed gastric mucosa with intestinal metaplasia and cytologic atypia but no appreciable tumor cells, ischemia, mucormycosis, or a possible malignancy (Figure 3). A repeat EGD with biopsy was done the next day (Figure 4). This time, histopathology demonstrated a poorly differentiated carcinoma without definitive squamous or glandular differentiation due to the scant tumor cells obtained (Figure 5 and Figure 6). A periodic acid-Schiff stain was negative for fungal organisms. There was no evidence of Helicobacter pylori (H. pylori), cytomegalovirus, herpes simplex virus, or Candida albicans infections. Tissue culture was also negative.
The patient was gradually introduced to oral intake and was discharged home after making a significant recovery after five days of hospitalization to see an oncologist in the clinic. Meanwhile, a positron emission tomography/computed tomography (PET/CT) was scheduled to be completed two days after hospital discharge. However, he returned to the hospital with acute hypoxic respiratory failure and passed away one week later.
Discussion
Black esophagus is a rare condition characterized by the diffuse circumferential black discoloration of the distal esophageal mucosa. Its incidence may be underestimated as most cases are detected incidentally following presentation with UGIB in the emergency ED [1]. Its peak incidence in older males correlates with the prevalence of multiple comorbidities, hence, its multifactorial association. A high index of suspicion for AEN is necessary, especially in elderly males with UGIB and hemodynamic compromise, as AEN is associated with high mortality rates.
Maintaining blood flow to the layers of the esophageal wall is vital for cellular function and protective barriers. A compromise of blood supply can lead to cell death and tissue necrosis. Conditions associated with hypotension (e.g., hypovolemia, low flow states, thromboembolic conditions, hemodialysis, and congestive heart failure), infection (e.g., Candida esophagitis, sepsis, septic shock), medication (e.g., chemotherapy), and failure to protect mucosal integrity (e.g., GERD, hiatal hernia, and esophageal varices), as well as solid tumors, hematological malignancies, antiphospholipid antibody syndrome, and atherosclerosis, have been linked to the development of AEN [5],[6]. Other associated conditions include T2DM, diabetic ketoacidosis, poorly controlled HTN, alcohol use disorder, chronic kidney disease, intoxication, cirrhosis, and smoking. Recognizing risk factors for developing black esophageal necrosis promptly could dramatically alter the disease course [3].
The distal third of the esophagus is most affected due to its relatively scant vascularization. Still, a proximal extension of mucosal injury has been reported, and in approximately 34% of cases, the entire esophagus may appear black [1].
Although not standardized, AEN is divided into four stages based on clinical presentation and endoscopic findings [1].
- Hyperemia and edema present with dysphagia, hematemesis, or melena.
- Necrosis characterized by severe epigastric pain, bleeding, and signs of systemic inflammation or shock.
- Ulceration and perforation are potentially life-threatening.
- Healing and fibrotic stage associated with dysphagia or odynophagia from esophageal stricture and granulation tissue, gradually replacing necrotic tissue, thus appearing less black.
Acute esophageal necrosis (AEN) can be incidentally found on endoscopy in asymptomatic patients. Still, typically, 90% present with signs of UGIB, manifesting as hematemesis or melena, with or without anemia or hemodynamic compromise. Other symptoms include dysphagia, acute abdominal pain (28%), and, in severe cases, fever with signs of sepsis and multiorgan failure [3]. Physical examination findings in most patients include but are not limited to, anemia, hypotension, tachycardia, or cachexia. Common laboratory findings include low hemoglobin, leukocytosis, and elevated lactate, which indicate tissue hypoperfusion [1].
Radiologically, esophageal wall thickening is the most consistent but non-specific finding. Other findings may include hiatal hernia, distended stomach, and mediastinal or subcutaneous gas in cases of perforation [3]. Endoscopically, AEN presents as a black esophagus of varying lengths with abrupt demarcation at the gastroesophageal junction (GEJ) [3]. Histological examination is crucial for diagnosing underlying pathology and differentiating AEN from other causes of melanocytic esophagus, such as melanoma, pseudomelanosis, mucormycosis, or other superimposed infections [1].
In our case, chronic injury from untreated GERD possibly gave rise to Barrett’s esophagus with intestinal metaplasia, resulting in mucosal and vascular disruption of the distal esophagus. Contributing factors included poorly controlled diabetes mellitus, hypertension, history of smoking, and poor hospital follow-up. Despite the association of AEN with alcohol, our patient drank sparingly. One cannot, therefore, quantify the contribution of an individual risk factor to disease pathogenesis. He presented with massive hematemesis, a Hb of 10.7 g/dL, and a white blood cell (WBC) of 18.2 cells/L, indicating inflammation from necrosis. Computed tomography (CT) scan revealed a circumferentially thickened esophagus with necrotic debris and evidence of metastasis. Esophagogastroduodenoscopy confirmed the black esophagus with marked demarcation at the GEJ (Figure 5).
Obtaining histological samples was challenging due to the friability of the esophagus, which increases the risk of perforation. Initial histology was non-diagnostic, showing gastritis with intestinal metaplasia and cytologic atypia but no tumor cells, H. pylori, or fungi. A repeat biopsy eventually revealed scant tumor cells despite clinical and radiological evidence of metastasis, raising concerns for under-diagnosis of cancer in AEN patients.
Management of acute esophageal necrosis (AEN) involves immediate stabilization with fluid resuscitation, IV proton pump inhibitors, blood transfusion, and correction of electrolyte imbalances. Supportive care, including antiemetics, antipyretics, and analgesics, is also necessary. Nutritional support through non-oral routes is crucial to esophageal healing. Treating the underlying cause is essential, involving appropriate staging with PET/CT scans, prompt hematology referral, and initiation of curative or palliative chemotherapy or radiotherapy
Despite its association with inflammation and leukocytosis, there are no current guidelines for using antibiotics in AEN. Our patient had a heart rate of 140 bpm, respiratory rate of 25 breaths/min, and WBC of 18 cells/L, meeting sepsis criteria; these improved with resuscitation without antibiotics, raising questions about the impact of antibiotic use on mortality. More research is needed to guide antibiotic use in AEN patients.
Addressing complications, such as strictures, with endoscopic dilatation is often required, and surgical intervention may be necessary in cases of perforation. Prompt referral to an oncologist is critical for survival. In our case, the patient returned to the ED with acute respiratory failure and died a few hours after, before oncologist evaluation, despite a good recovery on initial hospitalization.
Conclusion
Given the multifactorial etiology of AEN, clinical dilemmas in identifying and mitigating rarer causes are expected. This case highlights the consequences of poor follow-up care once a risk factor is determined, with long-standing GERD leading to Barrett’s esophagus with metaplasia, progressing to distal esophageal cancer, and death from sequelae of AEN.
REFERENCES
1.
Dias E, Santos-Antunes J, Macedo G. Diagnosis and management of acute esophageal necrosis. Ann Gastroenterol 2019;32(6):529–40. [CrossRef]
[Pubmed]

2.
Goldenberg SP, Wain SL, Marignani P. Acute necrotizing esophagitis. Gastroenterology 1990;98(2):493–6. [CrossRef]
[Pubmed]

3.
Siddiqi A, Chaudhary FS, Naqvi HA, Saleh N, Farooqi R, Yousaf MN. Black esophagus: A syndrome of acute esophageal necrosis associated with active alcohol drinking. BMJ Open Gastroenterol 2020;7(1):e000466. [CrossRef]
[Pubmed]

4.
Gurvits GE, Shapsis A, Lau N, Gualtieri N, Robilotti JG. Acute esophageal necrosis: A rare syndrome. J Gastroenterol 2007;42(1):29–38. [CrossRef]
[Pubmed]

5.
Gurvits GE. Black esophagus: Acute esophageal necrosis syndrome. World J Gastroenterol 2010;16(26):3219–25. [CrossRef]
[Pubmed]

6.
Samuel S, Passarella A, Tsyrlin R, Hasak S, Gurvits G. Acute esophageal necrosis in a patient with severe cardiovascular disease and arrhythmia. Cureus 2024;16(7):e65691. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Acknowledgments
We sincerely thank the clinicians who managed this patient at West Virginia University Hospital.
Author ContributionsOlanrewaju Adeniran - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Farirai Marwizi - Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Joshua Kirkpatrick - Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ayowumi Adekolu - Conception of the work, Design of the work, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Abdullahi Sulaiman - Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ethan M Cohen - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Mohamed Zitun - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Olanrewaju Adeniran et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.