|
Case Report
Early diagnosis of Legionnaires’ pneumonia in an immunocompromised neuroblastoma patient using metagenomic next-generation sequencing: A case report
1 Department of Clinical Laboratory, Center for Laboratory Medicine, Hainan Women and Children’s Medical Center, Hainan Medical University, Haikou, Hainan 570206, China
2 Department of Microbiology, Faculty of Medicine-Lincoln University College, Petaling Jaya, Malaysia
3 Hainan Medical University-The University of Hong Kong Joint Laboratory of Tropical Infectious Diseases, Key Laboratory of Tropical Translational Medicine of Ministry of Education, School of Basic Medicine and Life Sciences, Hainan Medical University, Haikou, Hainan 571199, China
4 Respiratory Department, Northumbria Health Care NHS Foundation Trust, Care of Gail Hewitt, Newcastle NE23 6NZ, UK
5 Department of Pathogen Biology, Hainan Medical University, Haikou 571199, China
Address correspondence to:
Meng Chang
Department of Clinical Laboratory, Center for Laboratory Medicine, Hainan Women and Children’s Medical Center, Hainan Medical University, Haikou, Hainan 570206,
China
Message to Corresponding Author
Article ID: 101478Z01MX2024
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Xiao M, Banu A, Zeng X, Shi S, Chen S, Ge N, Tang C, Huang Y, Wang G, Hu X, Cui X, Yin F, Peng R, Chang M. Early diagnosis of Legionnaires’ pneumonia in an immunocompromised neuroblastoma patient using metagenomic next-generation sequencing: A case report. Int J Case Rep Images 2024;15(2):89–95.ABSTRACT
Introduction: Legionella pneumophila (L. pneumophila) is an aerobic, Gram-negative intracellular pathogen commonly responsible for community-acquired pneumonia and a significant causative agent in hospital-acquired pneumonia. Immunocompromised individuals, such as those undergoing organ transplantation, chemotherapy, or corticosteroid therapy, are particularly vulnerable to L. pneumophila infections. These infections often lead to Legionnaires’ pneumonia, which is challenging to differentiate from other respiratory pathogen infections based solely on clinical presentation. Moreover, traditional pathogen detection methods have low sensitivity for Legionella, further complicating the diagnosis of Legionnaires’ pneumonia.
Case Report: This case report presents a 6-year-and-9-month-old boy who developed persistent high fever during chemotherapy following the surgical resection of a retroperitoneal neuroblastoma. Imaging studies revealed significant pneumonia manifestations. Metagenomic next-generation sequencing (mNGS) of nasopharyngeal swabs and bronchoalveolar lavage fluid confirmed the diagnosis of Legionnaires’ pneumonia.
Conclusion: This case highlights the challenges and critical importance of early and accurate diagnosis of Legionella infections, particularly in immunocompromised patients such as those undergoing chemotherapy. The use of advanced diagnostic techniques like metagenomic next-generation sequencing (mNGS) is essential for the prompt identification and treatment of Legionnaires’ pneumonia in this vulnerable population, improving patient outcomes. This report emphasizes the need for heightened awareness and advanced diagnostic techniques in managing atypical pneumonia pathogens in vulnerable populations.
Keywords: Immunocompromised patient, Legionella pneumophila, Legionnaires pneumonia, Metagenomic next-generation sequencing
Introduction
Legionella pneumophila (L. pneumophila), an intracellular bacterium prevalent in various environments, is classified as an opportunistic pathogen [1],[2],[3]. This Gram-negative, aerobic organism was named following an outbreak of an unexplained respiratory illness identified in Philadelphia, USA, in 1976 [4]. Its genome is comprised of a circular chromosome roughly 3.3 Mb in size [5]. Legionella pneumophila is a significant cause of both community-acquired and nosocomial pneumonia, particularly in immunocompromised individuals [6]. Reports indicate that at least 150 outbreaks of legionellosis attributable to L. pneumophila have occurred worldwide, with immunocompromised populations being at heightened risk [7]. Legionella pneumophila infections account for up to 10% of all bacterial pneumonias, with mortality rates significantly higher in immunocompromised patients, including those undergoing chemotherapy [8]. Legionnaires’ disease (LD) is classified as an atypical pneumonia, with clinical manifestations that often include extrapulmonary symptoms such as gastrointestinal and neurological issues, which can complicate early diagnosis compared to typical bacterial pneumonias [9].
Legionella pneumophila is extensively found in natural water systems, including freshwater reservoirs and waterways, as well as in artificial water systems such as landscape fountains, plumbing systems, air conditioning units, and shower installations [10],[11],[12]. Legionella pneumophila thrives in warm water environments, with optimal growth occurring between 20 and 42°C. This characteristic enables the bacterium to persist in artificial water systems such as air conditioning units, plumbing, and other warm water sources [9]. The ability of L. pneumophila to proliferate within biofilms offers it protection against environmental stresses like disinfection [13]. Human infection ensues following the inhalation or aspiration of aerosols harboring the pathogen [14]. Upon infection, L. pneumophila invades and replicates within alveolar macrophages, mirroring its infection of protozoan hosts in the environment [15]. The bacterium’s virulence is attributed to factors such as flagella, fimbriae, type II and type IV secretion systems, and iron-acquisition strategies [16]. Among the more than 15 identified serogroups, L. pneumophila serogroup 1 is the primary causative agent of legionellosis [7].
As a significant respiratory pathogen, L. pneumophila infections can vary from mild symptoms such as fever, muscle aches, and nausea, to severe manifestations such as respiratory distress, respiratory failure, multi-organ failure, and potentially death [17]. Immunocompromised individuals, such as those undergoing chemotherapy, are at higher risk for severe L. pneumophila infections. These infections can progress rapidly, leading to severe pneumonia, multi-organ failure, and septic shock [18]. Extrapulmonary manifestations such as neurological or gastrointestinal symptoms may also occur, often leading to early misdiagnosis. Additionally, the bacterium requires a specialized medium for culturing, complicating the diagnostic process in the absence of positive microbiological tests. Previous reports of L. pneumophila infections have typically been confirmed using polymerase chain reaction (PCR) targeting specific microorganisms rather than through next-generation sequencing (NGS) [19],[20],[21]. Next-generation sequencing offers a broader, unbiased diagnostic approach, particularly useful in immunocompromised patients presenting with atypical pneumonia symptoms.
Immunocompromised individuals, including cancer patients undergoing chemotherapy, are at increased risk of severe infections [22]. Neuroblastoma, a malignant tumor arising from neural crest cells, often necessitates intensive chemotherapy, resulting in prolonged immunosuppression [23]. This report outlines the clinical progression, diagnostic challenges, and management of a pediatric neuroblastoma patient who developed LD during chemotherapy-induced bone marrow suppression. The case highlights the critical need to address L. pneumophila infections in immunocompromised individuals, where traditional diagnostic methods may be inadequate. Advanced diagnostic techniques, such as mNGS, play a pivotal role in the early detection of these infections, allowing for timely intervention and better control of lower respiratory tract infections in this vulnerable population.
Case Report
A 6-year-and-9-month-old boy was admitted on August 6, 2023, for subsequent chemotherapy due to bilateral lower limb pain and fever persisting for three days. This occurred six months post-operation for a malignant retroperitoneal tumor. The patient was diagnosed with stage IV retroperitoneal neuroblastoma (INSS IV) in July 2022. Following surgery on December 30, 2022, the patient underwent multiple cycles of chemotherapy and radiotherapy, with the most recent chemotherapy session administered on July 12, 2023.
Upon admission, physical examination revealed a temperature of 38.4°C, pulse rate of 120 beats per minute, respiratory rate of 24 breaths per minute, and blood pressure of 98/55 mmHg. The patient measured 111 cm in height and weighed 16 kg. There were no rashes or palpable lymphadenopathy, and respiratory examination showed clear lung sounds bilaterally. Abdominal examination noted a 30 cm surgical scar in the upper abdomen, with normal bowel sounds. Neurological examination was unremarkable, and peripheral circulation was intact. The clinical parameters observed during the patient’s hospitalization are presented in Figure 1.
Initial laboratory investigations showed a white blood cell count of 7.3 × 109/L with 83.5% neutrophils, hemoglobin level of 79 g/L, platelet count of 51 × 109/L, C-reactive protein (CRP) level of 124.83 mg/L, and serum amyloid A >320 mg/L. On the same day (August 6, 2023), imaging (DR examination) revealed that both lungs exhibited prominent markings, and soft tissue shadow in the lower mediastinum comparable to previous findings. The patient was treated with cefoperazone-sulbactam for suspected bacterial infection. Despite treatment, the patient continued to experience pain and recurrent fevers over the next two days. Given the possibility of tumor-induced fever, on August 8, 2023, the patient resumed a high-risk neuroblastoma chemotherapy regimen, which included cyclophosphamide, doxorubicin, and vincristine.
Subsequent laboratory tests on August 11, 2023, showed worsening bone marrow suppression with a white blood cell count of 1.7 × 109/L, hemoglobin level of 68 g/L, platelet count of 36 × 109/L, and CRP level of 213.90 mg/L. The patient received granulocyte colony-stimulating factor (G-CSF) and red blood cell transfusions.
Despite initial improvement, the patient developed high-grade fevers and worsening respiratory symptoms on August 21, 2023. Repeat chest imaging on August 22, 2023, identified new lung nodules and consolidation, suggesting secondary infection. Blood cultures grew Streptococcus mitis/oralis, prompting a switch to imipenem–cilastatin therapy. Metagenomic next-generation sequencing of respiratory samples confirmed L. pneumophila infection.
Detailed Progression and Management
Day 1 (on August 6, 2023): Initial imaging studies included a chest X-ray which showed increased lung markings but no clear consolidation or pleural effusion. Laboratory findings revealed elevated inflammatory markers and a neutrophilic predominance. On the same day, the patient was empirically started on cefoperazone-sulbactam, considering his immunocompromised state and the risk of bacterial infection.
Day 3: The patient’s condition did not improve with initial antibiotic therapy. Given the possibility of tumor-related fever, chemotherapy was resumed on Day 3, following the high-risk neuroblastoma protocol. The regimen included cyclophosphamide (1380 mg, days 0–1), doxorubicin (16.5 mg, days 0–2), and vincristine (0.44 mg, days 0–2). Despite chemotherapy, the patient continued to exhibit high fevers and worsening clinical status.
Day 6: Repeat blood tests indicated significant bone marrow suppression with a white blood cell count of 1.7 × 109/L, hemoglobin level of 68 g/L, and platelet count of 36 × 109/L. The elevated CRP level (213.90 mg/L) suggested ongoing systemic inflammation. Given the patient’s critical state, G-CSF was administered to stimulate white blood cell production, and red blood cell transfusions were given to manage anemia. This sequence follows chemotherapy-related events.
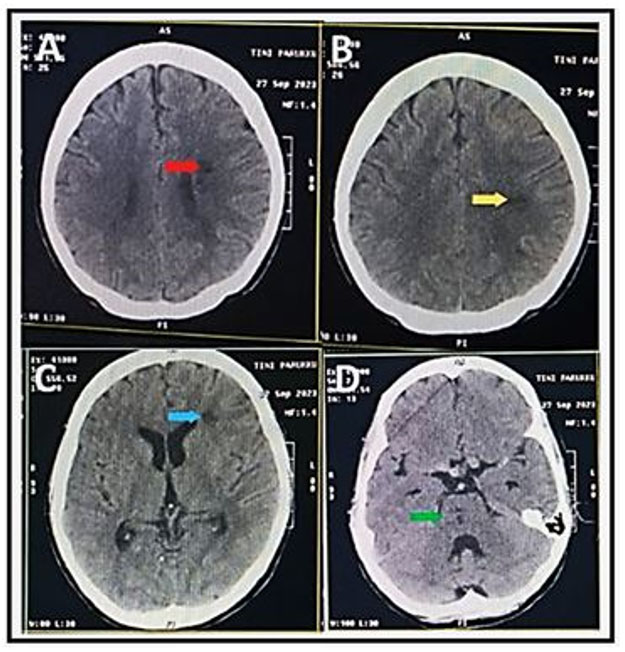
Day 16: The patient experienced recurrent fevers with temperatures reaching 39.3°C. Given the failure to respond to initial antibiotic therapy and the persistent fever, the clinical team considered a broader differential diagnosis, including atypical pathogens. On Day 17, chest computed tomography (CT) revealed new pulmonary nodules and areas of consolidation suggestive of secondary infection (Figure 2).
Further microbiological investigations, including blood cultures, identified Streptococcus mitis/oralis. Antibiotic therapy was escalated to imipenem-cilastatin, given the identification of this organism and its potential for causing significant infections in immunocompromised hosts. Despite aggressive antibiotic therapy, the patient’s condition continued to deteriorate. The persistent fever and progressive respiratory symptoms necessitated the use of advanced diagnostic techniques. On Day 18, mNGS of nasopharyngeal swabs and bronchoalveolar lavage fluid was performed, revealing the presence of L. pneumophila DNA.
Upon identification of L. pneumophila, antibiotic therapy was adjusted to include vancomycin and levofloxacin. The decision to use levofloxacin was based on its effectiveness against Legionella spp. Additionally, trimethoprim-sulfamethoxazole was added to broaden the coverage against possible co-infections.
Day 19: The patient exhibited signs of severe respiratory distress, including tachypnea, retractions, and hypoxemia. Non-invasive mechanical ventilation was initiated, but the patient’s respiratory status continued to worsen, necessitating intubation and mechanical ventilation. Supportive care included the administration of midazolam and fentanyl for sedation and pain management.
Despite maximal supportive care, the patient’s condition continued to decline. On Day 20, repeat chest X-ray indicated worsening bilateral pulmonary infiltrates and right-sided pleural effusion. Laboratory tests showed persistent leukopenia (white blood cell count of 0.1 × 109/L), severe anemia (hemoglobin level of 71 g/L), and thrombocytopenia (platelet count of 17 × 109/L). C-reactive protein remained markedly elevated at 229.49 mg/L.
Given the ongoing critical illness and poor response to therapy, the patient was diagnosed with sepsis and septic shock. Intensive care management included the use of norepinephrine to maintain blood pressure, albumin infusion to manage hypoalbuminemia, and continuous monitoring for signs of organ dysfunction.
Day 21: Despite aggressive interventions, the patient’s condition remained critical. His family was informed of the grave prognosis and the likely outcome. After thorough discussions and considering the patient’s critical state, the family opted to withdraw life-sustaining treatments and the patient was discharged home with hospice care.
Discussion
This case highlights the critical role of advanced diagnostic techniques, particularly mNGS [24], in managing severe infections in immunocompromised pediatric patients. Legionella pneumophila, while rare, can lead to severe respiratory infections in this vulnerable population, as evidenced by the patient’s presentation and clinical course. The rapid progression of the disease in this case underscores the challenges in managing infections in immunocompromised patients, where symptoms may overlap with other conditions, making diagnosis and treatment more complex.
While the severity of the disease and the extent of inflammation linked to Legionella are yet to be fully established, this case illustrates the importance of recognizing co-infections in immunosuppressed individuals [25]. Broader awareness of atypical pathogens, including L. pneumophila, is essential for clinicians treating patients with compromised immune systems, such as those undergoing chemotherapy. In this case, L. pneumophila infection was complicated by the patient’s prolonged immunosuppression due to intensive chemotherapy for neuroblastoma.
Neuroblastoma and its aggressive treatment regimen, including chemotherapy and radiotherapy, often result in profound immunosuppression, which significantly increases the risk of severe infections [26]. Complications of neuroblastoma, such as bone marrow suppression, can exacerbate the vulnerability of pediatric patients to opportunistic pathogens like L. pneumophila. Chemotherapy-induced neutropenia, thrombocytopenia, and mucosal barrier disruption further complicate the management of infections, as they limit the body’s ability to mount an effective immune response. In this case, the patient’s immune system was heavily compromised, likely contributing to the progression and severity of the infection [27].
With advancements in technology, mNGS plays a pivotal role in identifying pathogens that may not be easily detected using traditional methods. While mNGS was critical in this case for identifying the atypical pathogen, timely clinical decision-making and the prompt initiation of appropriate therapy are equally important [28]. This case emphasizes that mNGS is a powerful tool, but it is most effective when used in conjunction with other diagnostic and therapeutic interventions.
In terms of treatment, the decision to use levofloxacin was based on its known efficacy against Legionella spp., but the patient’s severely compromised immune system likely hindered the effective clearance of the pathogen, despite early identification. This reflects the broader issue in managing infections in pediatric oncology patients, where both the underlying malignancy and the aggressive treatment regimens contribute to an increased risk of severe infections [29].
Additionally, this case demonstrates the challenges clinicians face when families opt to withdraw life-sustaining treatment, despite the potential for recovery. In this instance, the child’s family decided to discharge the patient on the second day of targeted therapy, following discussions about the grave prognosis.
The management of infections in immunocompromised patients is challenging due to the broad differential diagnoses and the need for timely, accurate diagnosis. While traditional diagnostic methods, such as culture and serology, may be useful, they often take longer and may not yield results in patients who have already received antibiotics. The combination of clinical vigilance and rapid diagnostics, such as mNGS, allows for a more tailored approach to treatment.
In this case, it underscores the necessity of an integrated approach to managing severe infections in immunocompromised patients. It highlights the importance of advanced diagnostics, but also the need for clinical judgment, patient management, and the role of timely therapeutic interventions to improve outcomes.
Conclusion
In conclusion, this case report highlights the pivotal role of mNGS in diagnosing LD in immunocompromised pediatric patients, such as those undergoing chemotherapy for neuroblastoma. The patient, who presented with severe infection and persistent symptoms despite conventional treatment, was accurately diagnosed with L. pneumophila infection through mNGS, underscoring the limitations of traditional diagnostic methods in such complex cases. The rapid identification of the pathogen facilitated timely and appropriate antimicrobial therapy, demonstrating the clinical utility of mNGS in managing atypical infections in vulnerable populations. This case underscores the necessity for advanced diagnostic tools and vigilant pathogen surveillance to improve outcomes in immunosuppressed patients, advocating for their integration into routine clinical practice to enhance the detection and management of severe infections.
REFERENCES
1.
Nisar MA, Ross KE, Brown MH, Bentham R, Whiley H. Legionella pneumophila and protozoan hosts: Implications for the control of hospital and potable water systems. Pathogens 2020;9(4):286. [CrossRef]
[Pubmed]

2.
Khaledi A, Esmaeili SA, Vazini H, Karami P, Bahrami A, Sahebkar A. Evaluation of the prevalence of Legionella pneumophila in Iranian clinical samples: A systematic review and meta-analysis. Microb Pathog 2019:129:93–8. [CrossRef]
[Pubmed]

3.
Miyashita N, Higa F, Aoki Y, et al. Distribution of Legionella species and serogroups in patients with culture-confirmed Legionella pneumonia. J Infect Chemother 2020;26(5):411–7. [CrossRef]
[Pubmed]

4.
Fraser DW, Tsai TR, Orenstein W, et al. Legionnaires' disease: Description of an epidemic of pneumonia. N Engl J Med 1977;297(22):1189–97. [CrossRef]
[Pubmed]

5.
Mercante JW, Winchell JM. Current and emerging Legionella diagnostics for laboratory and outbreak investigations. Clin Microbiol Rev 2015;28(1):95–133. [CrossRef]
[Pubmed]

6.
Rello J, Allam C, Ruiz-Spinelli A, Jarraud S. Severe Legionnaires’ disease. Ann Intensive Care 2024;14(1):51. [CrossRef]
[Pubmed]

7.
Gonçalves IG, Simões LC, Simões M. Legionella pneumophila. Trends Microbiol 2021;29(9):860–1. [CrossRef]
[Pubmed]

8.
Fiumefreddo R, Zaborsky R, Haeuptle J, et al. Clinical predictors for Legionella in patients presenting with community-acquired pneumonia to the emergency department. BMC Pulm Med 2009;9:4. [CrossRef]
[Pubmed]

9.
Fields BS, Benson RF, Besser RE. Legionella and Legionnaires’ disease: 25 years of investigation. Clin Microbiol Rev 2002;15(3):506–26. [CrossRef]
[Pubmed]

10.
Barker KA, Whitney EA, Blake S, Berkelman RL. A review of guidelines for the primary prevention of Legionellosis in long-term care facilities. J Am Med Dir Assoc 2015;16(10):832–6. [CrossRef]
[Pubmed]

11.
Barrabeig I, Rovira A, Garcia M, et al. Outbreak of Legionnaire’ disease associated with a supermarket mist machine. Epidemiol Infect 2010;138(12):1823–8. [CrossRef]
[Pubmed]

12.
Cowen KA, Ollison WM. Continuous monitoring of particle emissions during showering. J Air Waste Manag Assoc 2006;56(12):1662–8. [CrossRef]
[Pubmed]

13.
Piao Z, Sze CC, Barysheva O, Iida K, Yoshida S. Temperature-regulated formation of mycelial mat-like biofilms by Legionella pneumophila. Appl Environ Microbiol 2006;72(2):1613–22. [CrossRef]
[Pubmed]

14.
Diederen BMW. Legionella spp. and Legionnaires’ disease. J Infect 2008;56(1):1–12. [CrossRef]
[Pubmed]

15.
Franco IS, Shohdy N, Shuman HA. The Legionella pneumophila effector VipA is an actin nucleator that alters host cell organelle trafficking. PLoS Pathog 2012;8(2):e1002546. [CrossRef]
[Pubmed]

16.
Shames SR, Liu L, Havey JC, Schofield WB, Goodman AL, Roy CR. Multiple Legionella pneumophila effector virulence phenotypes revealed through high-throughput analysis of targeted mutant libraries. Proc Natl Acad Sci U S A 2017;114(48):E10446–54. [CrossRef]
[Pubmed]

17.
Cunha BA, Burillo A, Bouza E. Legionnaires’ disease. Lancet 2016;387(10016):376–85. [CrossRef]
[Pubmed]

18.
Phin N, Parry-Ford F, Harrison T, et al. Epidemiology and clinical management of Legionnaires’ disease. Lancet Infect Dis 2014;14(10):1011–21. [CrossRef]
[Pubmed]

19.
Zhang Q, Zhou H, Chen R, et al. Legionnaires’ disease caused by Legionella pneumophila serogroups 5 and 10, China. Emerg Infect Dis 2014;20(7):1242–3. [CrossRef]
[Pubmed]

20.
Buchholz U, Reber F, Lehfeld AS, et al. Probable reinfection with Legionella pneumophila – A case report. Int J Hyg Environ Health 2019;222(2):315–8. [CrossRef]
[Pubmed]

21.
Du R, Feng Y, Wang Y, Huang J, Tao Y, Mao H. Metagenomic next-generation sequencing confirms the diagnosis of Legionella pneumonia with rhabdomyolysis and acute kidney injury in a limited resource area: A case report and review. Front Public Health 2023:11:1145733. [CrossRef]
[Pubmed]

22.
Zembower TR. Epidemiology of infections in cancer patients. Cancer Treat Res 2014:161:43–89. [CrossRef]
[Pubmed]

23.
Ponzoni M, Bachetti T, Corrias MV, et al. Recent advances in the developmental origin of neuroblastoma: An overview. J Exp Clin Cancer Res 2022;41(1):92. [CrossRef]
[Pubmed]

24.
Yi H, Fang J, Huang J, Liu B, Qu J, Zhou M. Legionella pneumophila as cuse of severe community-acquired Pneumonia, China. Emerg Infect Dis 2020;26(1):160–2. [CrossRef]
[Pubmed]

25.
Head BM, Trajtman A, Bernard K, et al. Legionella co-infection in HIV-associated pneumonia. Diagn Microbiol Infect Dis 2019;95(1):71–6. [CrossRef]
[Pubmed]

26.
Whittle SB, Smith V, Doherty E, Zhao S, McCarty S, Zage PE. Overview and recent advances in the treatment of neuroblastoma. Expert Rev Anticancer Ther 2017;17(4):369–86. [CrossRef]
[Pubmed]

27.
Whittle SB, Williamson KC, Russell HV. Incidence and risk factors of bacterial and fungal infection during induction chemotherapy for high-risk neuroblastoma. Pediatr Hematol Oncol 2017;34(5):331–42. [CrossRef]
[Pubmed]

28.
Naccache SN, Federman S, Veeraraghavan N, et al. A cloud-compatible bioinformatics pipeline for ultrarapid pathogen identification from next-generation sequencing of clinical samples. Genome Res 2014;24(7):1180–92. [CrossRef]
[Pubmed]

29.
Wang X, Guo Y, Zhang Y, et al. Legionella pneumophila subspecies fraseri infection after allogeneic hematopoietic stem cell transplant, China. Emerg Infect Dis 2022;28(4):903–5. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Meifang Xiao - Conception of the work, Design of the work, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Afreen Banu - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Xiangyue Zeng - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Shengjie Shi - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Siqi Chen - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Nan Ge - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Cheng Tang - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Yi Huang - Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Gaoyu Wang - Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Xiaoyuan Hu - Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Xiuji Cui - Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Feifei Yin - Conception of the work, Design of the work, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ruoyan Peng - Conception of the work, Design of the work, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Meng Chang - Conception of the work, Design of the work, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2024 Meifang Xiao et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.