|
Clinical Image
Brain relapse of a mucosal melanoma
1 MD, Radiology Department, Oncology National Institute, Rabat, Morocco
2 Professor, Radiology Department, Oncology National Institute, Rabat, Morocco
Address correspondence to:
Amine Naggar
37, Lot Sanabil, Route Mehdia, Salé,
Morocco
Message to Corresponding Author
Article ID: 101351Z01AN2022
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Naggar A, Mohamed DA, Omor Y, Latib R. Brain relapse of a mucosal melanoma. Int J Case Rep Images 2022;13(2):160–163.ABSTRACT
No Abstract
Keywords: Brain, Melanoma, Metastases, MRI
Case Report
We report a case of a 52-year-old patient, with a history of a surgically cured locally advanced anorectal melanoma with pelvic lymphadenopathies. The diagnosis was based on histology: On a macroscopical level, it was a tumor located in the anorectal margin, exophytic, blackish, and friable. Microscopy showed an ulcerated mucosa, a chorion that is massively infiltrated by a high-density proliferation made of atypical round, oval, or elongated cells with atypical hyperchromatic nuclei, often nucleolated, with the presence, in areas, of melanic pigments. Immunohistochemistry showed a diffuse cytoplasmic signal on anti-HMB45 immunomarking.
No metastases were found on the initial workup, which included a computed tomography (CT) scan of the chest-abdomen-pelvis, a brain magnetic resonance imaging (MRI), and a positron emission tomography scan. And the treatment consisted of an abdomino-perineal amputation with a pseudo-continent perineal colostomy, followed by pembrolizumab immunotherapy.
The patient had been under control for more than a year, before presenting to the emergency room of our structure, for an acute setting of a right facial palsy and a blurred vision.
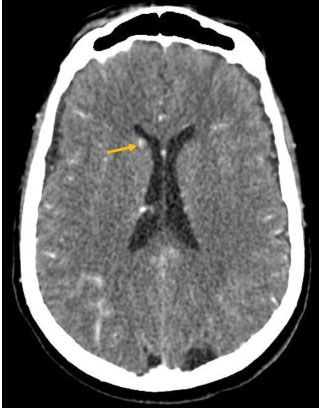
A brain CT was performed, showing multiple supra and infratentorial nodular lesions, homogeneously enhanced after contrast (Figure 1 and Figure 2).
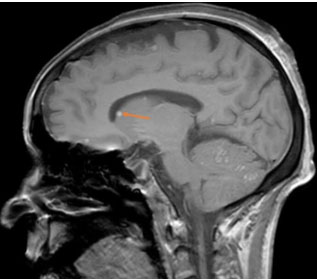
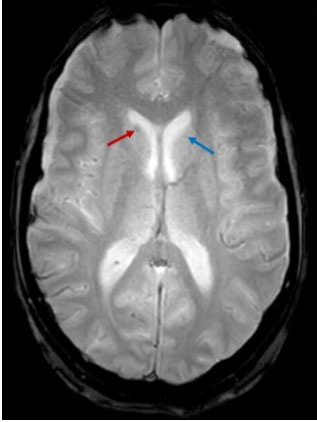
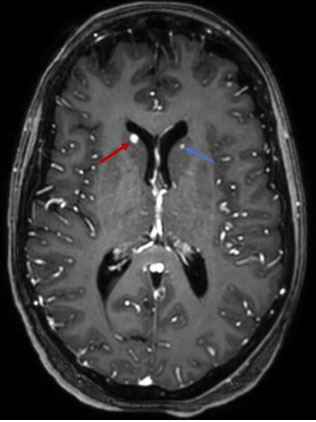
Brain MRI showed multiple nodular lesions in the frontal, parietal, occipital, and temporal lobes, in addition to the cerebellar hemispheres, not exceeding 2 cm in size, hyperintense on T1, hypointense on T2 and T2*, with nodular and ring-like enhancement after contrast (Figure 3, Figure 4, Figure 5), suggesting a melanomatous origin, in favor of a cerebral relapse of the anorectal melanoma.
We suggested stereotactic radiosurgery alone as treatment, since all the lesions were smaller than 2 cm. The patient was unfortunately lost to follow-up afterward.
Discussion
Extracutaneous malignant melanomas are rare tumors with a poor prognosis. Only 4–5% of all primary melanomas are noncutaneous [1].
Anorectal melanomas are rare entities accounting for 0.22% of all melanomas, 4.5% of noncutaneous melanomas, and 16.5% of mucosal melanomas [2]. It comprises 0.5–5% of all anorectal malignancies [3].
Melanomas are the second or third most common cause of cerebral metastases after lung and breast cancer, with a risk corresponding to 6.9% [4],[5],[6]. Cerebral metastases are the most ominous feature of malignant melanoma [7].
The mean time interval between the initial diagnosis of melanoma and appearance of intra-cranial metastases is estimated at 2.5 years, and the mean time interval between the initial diagnosis and development of the first symptoms of central nervous system (CNS) metastases is 3.5 years [8]. It corresponds to one year time interval in our case.
Symptoms and signs of cerebral metastases depend on the location of the tumor and its extent. There is no symptom that is specific for an intracranial metastases. Any recently appearing neurological symptom must suggest the diagnosis. Seizures, however, are highly suspicious since melanoma is one of the primary tumors that most commonly cause seizures. Headache and mental confusion, in the other hand, are the most common symptoms of brain metastases, in addition to motor dysfunction and nerve disturbance [9].
Radiological characteristics of melanomatous metastases are distinctive due to the presence of melanin and bleeding tendencies [10].
Computed tomography scan with and without contrast can detect most metastases above 10 mm and hemorrhagic lesions [11]. Metastatic lesions found on melanoma patients can be multiple or solitary. They are frequently located in the cerebral hemispheres, usually in the frontal lobes.
They often appear spontaneously hyperdense (75%), hypodense (22%), or isodense (3%), with post-contrast homogeneous nodular enhancement or ring-like pattern [8].
Magnetic resonance imaging is far more sensitive, especially for smaller lesions, lesions in the posterior cerebral fossa, and for leptomeningeal involvement [11]:
On T1-weighted imaging (WI): Half of melanomatous metastases is hyperintense, which is distinctive, since other cerebral metastases are rarely hyperintense on T1. This hypersignal is due to the intrinsic presence of blood and melanin.
On T2 WI: They are typically hypointense.
On T2* WI: In 71% of patients, these lesions present whether a hyposignal on T2*, a hypersignal on T1, or both. Therefore, the association of T1 and T2* sequences can help suggest a melanomatous origin of intra-cranial lesions.
Post-contrast T1 WI: It is the Gold standard for the diagnosis of cerebral metastases. It demonstrates nodular or ring-like enhancement [8],[10].
Differential diagnoses include hemorrhagic metastases of non-melanomatous origin, such as choriocarcinoma, renal cell carcinoma, thyroid, lung and breast cancer. Note that lung and breast cancer have a higher overall prevalence; Hemorrhagic metastases are usually iso- or hypointense on T1, but may also demonstrate T1 signal hyperintensity, depending on the age of hemorrhage.
Primary high-grade glioma is a differential diagnosis to solitary metastases. No individual parameter can accurately distinguish the two, however, evaluation of peritumoral edema, if there is one, by spectroscopy, perfusion, and diffusion-weighted imaging (DWI), may help lead to the accurate diagnosis (e.g., an ADC in the peritumoral edema that is higher than 1.3×10–3mm2/s is more suggestive of a metastasis) [12].
In our case, we had multiple brain lesions with a history of melanoma, which made the diagnosis easier. No peri-tumoral edema nor abnormality on DWI was noted.
When it comes to treatment, recent data have shown improved intracranial response with the newer molecules of systemic therapies such as BRAF/mitogen-activated protein (MAP) kinase inhibitors and immunotherapy (anti-programed cell death protein 1, anti-cytotoxic T lymphocyte-associated protein 4).
The suggestion of local therapy (surgery and stereotactic radiosurgery) would depend on patient performance, neurological symptoms, location, size, and number of brain metastasis. The advantages of surgery include rapid relief of pressure effect, evacuation of hemorrhage, and the possibilities of biopsies for diagnosis and molecular studies to select appropriate systemic therapy. Combining surgery for larger and symptomatic lesions with stereotactic radiosurgery for smaller lesions is a common practice in patients with multiple brain metastases. Following resection, radiotherapy to resection cavity is recommended to eliminate micrometastases or residual metastases [13].
Conclusion
Malignant melanoma is an aggressive cancer and is a frequent cause of cerebral metastases. The latter demonstrates distinctive imaging features on brain MRI (hyperintense on T1 and hypointense on T2*). A relapse, especially intracranial, is possible even after years of control. The development of any recent neurological sign should suggest the diagnosis in patients with a history of melanoma. Systemic immunotherapy, surgery, stereotactic radiosurgery, and post-operative radiotherapy are all tools in the therapeutic arsenal to control intra-cranial metastases.
REFERENCES
1.
Hussein MR. Extracutaneous malignant melanomas. Cancer Invest 2008;26(5):516–34. [CrossRef]
[Pubmed]

2.
McLaughlin CC, Wu XC, Jemal A, Martin HJ, Roche LM, Chen VW. Incidence of noncutaneous melanomas in the U.S. Cancer 2005;103(5):1000–7. [CrossRef]
[Pubmed]

3.
Wong VK, Lubner MG, Menias CO, et al. Clinical and Imaging features of noncutaneous melanoma. AJR Am J Roentgenol 2017;208(5):942–59. [CrossRef]
[Pubmed]

4.
Barnholtz-Sloan JS, Sloan AE, Davis FG, Vigneau FD, Lai P, Sawaya RE. Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the Metropolitan Detroit Cancer Surveillance System. J Clin Oncol 2004;22(14):2865–72. [CrossRef]
[Pubmed]

5.
Bertolini F, Spallanzani A, Fontana A, Depenni R, Luppi G. Brain metastases: An overview. CNS Oncol 2015;4(1):37–46. [CrossRef]
[Pubmed]

6.
Amer MH, Al-Sarraf M, Baker LH, Vaitkevicius VK. Malignant melanoma and central nervous system metastases: Incidence, diagnosis, treatment and survival. Cancer 1978;42(2):660–8. [CrossRef]
[Pubmed]

7.
Douglas JG, Margolin K. The treatment of brain metastases from malignant melanoma. Semin Oncol 2002;29(5):518–24. [CrossRef]
[Pubmed]

8.
Goulart CR, Mattei TA, Ramina R. Cerebral melanoma metastases: A critical review on diagnostic methods and therapeutic options. ISRN Surg 2011;2011:276908. [CrossRef]
[Pubmed]

9.
Oberndorfer S, Schmal T, Lahrmann H, Urbanits S, Lindner K, Grisold W. The frequency of seizures in patients with primary brain tumors or cerebral metastases. An evaluation from the Ludwig Boltzmann Institute of Neuro-Oncology and the Department of Neurology, Kaiser Franz Josef Hospital, Vienna. [Article in German]. Wien Klin Wochenschr 2002;114(21–22):911–6.
[Pubmed]

10.
Gaviani P, Mullins ME, Braga TA, et al. Improved detection of metastatic melanoma by T2*-weighted imaging. AJNR Am J Neuroradiol 2006;27(3):605–8.
[Pubmed]

11.
Sloan AE, Nock CJ, Einstein DB. Diagnosis and treatment of melanoma brain metastasis: A literature review. Cancer Control 2009;16(3):248–55. [CrossRef]
[Pubmed]

12.
Fink KR, Fink JR. Imaging of brain metastases. Surg Neurol Int 2013;4(Suppl 4):S209–19. [CrossRef]
[Pubmed]

13.
Rishi A, Yu HM. Current treatment of melanoma brain metastasis. Curr Treat Options Oncol 2020;21(6):45. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Amine Naggar - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Daoud Ali Mohamed - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Youssef Omor - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Rachida Latib - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2022 Amine Naggar et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.