|

|
 |
|
Case Report
| ||||||
| Severe rhabdomyolysis induced renal failure after influenza vaccination in a patient on statins therapy | ||||||
| Shu-Hua Chen1, Cheng-Jui Lin2, Tung-Ying Chen3 | ||||||
|
1MD, Division of Nephrology, Department of Internal Medicine, Mackay Memorial Hospital, Taipei, Taiwan.
2MD, PhD, Division of Nephrology, Department of Internal Medicine, Mackay Memorial Hospital, 92 Chung San North Road, Section 2, Taipei 104, Taiwan. 3MD, Division of pathology, Department of Internal Medicine, Mackay Memorial Hospital, Taipei, Taiwan. | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] 
|
| How to cite this article |
| Shu-Hua C, Cheng-Jui L, Tung-Ying C. Severe rhabdomyolysis induced renal failure in a patient after an influenza vaccination. Int J Case Rep Images 2017;8(3):205–208. |
|
Abstract
|
|
Introduction:
There are various causes leading to acute rhabdomyolysis, and the most common potentially lethal complication of rhabdomyolysis is acute kidney injury. Influenza vaccination-associated rhabdomyolysis with acute renal failure is rarely reported in literature. There are few data regarding kidney biopsy results from rhabdomyolysis induced renal failure after influenza vaccination.
Case Report: A 55-year-old male with chronic kidney disease stage 3a with hyperlipidemia under rosuvastatin control, who was admitted due to severe rhabdomyolysis induced renal failure after influenza vaccination. Kidney biopsy revealed acute tubular necrosis (ATN) and acute interstitial nephritis (AIN) superimposed on focal segmental glomerulosclerosis. Conclusion: Our experience illustrates the dual pathologic findings of ATN and AIN, both induced by influenza vaccine in a patient with chronic kidney disease on statin therapy. | |
|
Keywords:
Acute tubular interstitial nephritis, Influenza vaccination, Kidney biopsy, Renal failure, Rhabdomyolysis
| |
|
Introduction
| ||||||
|
There are several causes of rhabdomyolysis, including trauma, exertion, lipid-lowering agents, infection, electrolyte imbalance and toxins. Injury of skeletal muscle leads to release of intramuscular contents, including creatine phosphokinase and myoglobin, often resulting in acute kidney injury. Acute kidney injury secondary to rhabdomyolysis is fairly common, representing about 13–50% of all cases of rhabdomyolysis [1] [2] [3]. There are few reports of influenza vaccination-induced rhabdomyolysis in patients under treatment with lipid-lowering agents. Our report presented a patient, whom had chronic kidney disease and hyperlipidemia under statin therapy, developed severe rhabdomyolysis after an influenza vaccination. | ||||||
|
Case Report
| ||||||
|
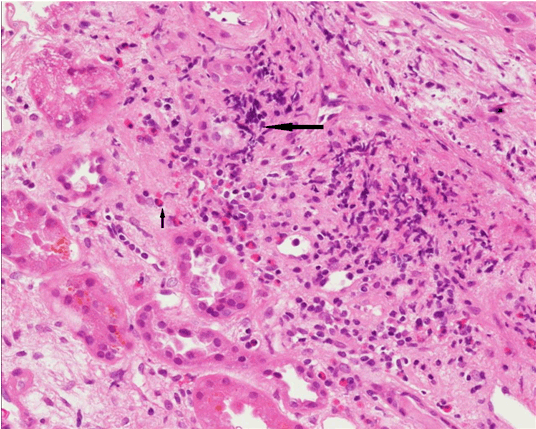
A 55-year-old male with history of chronic kidney disease stage 3a and hyperlipidemia with baseline creatinine 1.5 mg/dL two years ago. He was admitted to our hospital with symptoms of severe muscle pain and weakness. He took rosuvastatin 5 mg per day for hyperlipidemia for over one year. The patient received trivalent influenza vaccine (Sanofi Pasteur) five days prior to admission. He suffered from a single episode of fever and several times of diarrhea on the following four days. Subsequently, he developed general muscle soreness and weakness. Initial physical examination showed, blood pressure 91/61 mmHg, temperature 36°C, and pulse rate 84/min. Heart sounds were normal and the chest was clear to auscultation. He had dry mouth, poor skin turgor, dark urine, and tenderness of bilateral extremities, but normal muscle power. Laboratory data revealed hemoglobin 16.6 g/dL, platelets 214000/mm3, white blood cell count 23400/mm3, serum creatinine phosphokinase 17121 IU/L (normal levels <300), creatinine 3.5 mg/dL, blood urea nitrogen 36 mg/dL, aspartate aminotransferase 200 IU/L, alanine aminotransferase 69 IU/L, uric acid 13.2 mg/dL, sodium 136 mEq/L and potassium 4.0 mEq/L. Urine analysis disclosed high specific gravity 1.024, low pH value 5.0, protein 3+, trace glucosuria, trace ketone body, occult blood 3+, RBC 13 cells/HPF, WBC 6 cells/hpf. Daily urine protein loss was 2,413 mg/day. Other serologic tests: hepatitis B and C were negative; Anti-nuclear antibodies, p-ANCA, and c-ANCA were negative. Serum complement level was normal. After admission, rosuvastatin was discontinued. He was treated with aggressive intravenous fluid supplement and sodium bicarbonate alkalization therapy. Serum creatine kinase rose to a peak level of 77075 IU/L on the third day of admission (Figure 1). Urine myoglobin test was positive on the sixth day of admission. Concurrently, deterioration of renal function was noted (highest serum creatinine 12.2 mg/dL on 12th day of admission) and oliguria (daily urine amount < 200 ml) was noted as well (Figure 2). Temporary hemodialysis was total of six sessions between 3rd day and 14th day of admission. The level of serum creatine kinase and creatinine decreased gradually on the following days with intensive treatment. Renal biopsy was performed on the 17th day of admission, when daily urine amount was more than 2 liters. Histopathology, revealed the presence of advanced focal segmental glomerulosclerosis (FSGS) (Figure 3), acute tubular necrosis (ATN) (Figure 4) and allergy-associated acute interstitial nephritis (AIN) (Figure 5). Serum creatinine was 2.3 mg/dL on the 52th day. | ||||||
| ||||||
| ||||||
|
| ||||||
|
| ||||||
|
| ||||||
|
Discussion
| ||||||
|
Seasonal influenza vaccine contains inactivated viruses and is administered annually to protect against the influenza. According to Taiwan Centers for Disease Control and Prevention vaccination guidelines, influenza vaccines are recommended for everyone ages six months and older. Trivalent influenza vaccines protect against two influenza A strains, H1N1 and H3N2, and one influenza B strain. Mild side effects from the influenza vaccination include transient soreness, redness or swelling at the injection site, nausea, low-grade fever and headache. Rare but serious side effects were anaphylactic shock, difficult breathing, swelling around the eyes or lips, wheal, temporary thrombocytopenia, high fever and neurologic symptoms, such as Guillain–Barre syndrome [4]. In addition to lipid-lowering agents and virus infection, there are several reports about rhabdomyolysis triggered by seasonal influenza vaccination [5] [6] [7] [8][9]. Raman et al. found the first report of acute renal failure secondary to rhabdomyolysis following an influenza vaccination in a renal transplant recipient who had been on simvastatin and cyclosporine A therapy [5]. Novati et al. presented one case on statin therapy with acute renal failure twelve days after influenza vaccination, which consisted with acute tubulointerstitial nephropathy on urine analysis [9]. From our report, this patient developed severe rhabdomyolysis and acute allergic reactions with influenza vaccination. The renal pathological findings also demonstrated FSGS, ATN and AIN. The cause of rhabdomyolysis includes statin therapy as well as influenza vaccination. Statin may exert toxic effects on skeletal muscle, but whose overall incidence is typically <0.1% of patients receiving statin therapy [10]. Despite the widespread use of statin, the mechanism of statin-induced myopathy remains controversial and poorly understood. We know that, the ubiquitin-proteasome–dependent proteolytic pathway (UP pathway) is responsible for the breakdown of long-lived myofibrillar proteins (e.g., actin, myosin and troponin) in skeletal muscle, which releases of myoglobin. Statin may upregulate skeletal muscle gene expression of UP pathway [11]. Mechanisms of rhabdomyolysis induced acute kidney injury are renal vasoconstriction secondary to excessive leakage of extracellular fluid into the damaged muscle cells, formation of intratubular myoglobin casts that induce intra-tubular obstruction and direct toxin-related tubular damage [12]. In this case, no myoglobin cast was found in renal tubules on renal biopsy, which could be disappearing of myoglobin cast on the recovery of oliguria. Moreover, this patient had a previous history of chronic kidney disease (CKD) stage 3a, and was diagnosed as focal segmental glomerulosclerosis (FSGS) by kidney pathology. This can explain the patient's susceptibility for developing rhabdomyolysis induced renal failure after vaccination. Acute interstitial nephritis is often associated with use of drugs or infection, and is likely mediated through allergic mechanisms. In this patient, influenza vaccination may explain this hypersensitivity reaction. Although, influenza vaccination-associated rhabdomyolysis is rare, our report provides clinician a warning about serious complication in patients with renal impairment and statin therapy, that intend to be vaccinated against common viruses. | ||||||
|
Conclusion
| ||||||
|
In summary, rhabdomyolysis induced renal failure may contribute to the morbidity and mortality during hospitalization. We report a case developing severe rhabdomyolysis-induced renal failure with dual pathologic finding after an influenza vaccination. Kidney pathology demonstrates that acute tubular necrosis and acute interstitial nephritis may happen after an influenza vaccination. This report should caution clinicians about this potentially serious complication especially for those needed influenza vaccination with renal function impairment and on lipid-lowering medicine. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Shu-Hua Chen – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Cheng-Jui Lin – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Tung-Ying Chen – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2017 Shu-Hua Chen et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|