| Table of Contents |  |
|
Case Report
|
| Rapid diagnosis of sinonasal cancer in an anticoagulated patient: Outpatient clinical use of a gelatin-thrombin matrix (Floseal) for hemostasis following endonasal tumor biopsies |
| Shaun J Kilty1 |
|
1Associate, Department of Otolaryngology-Head & Neck Surgery, The Ottawa Hospital, Ottawa, Ontario, Canada.
|
|
doi:10.5348/ijcri-2011-11-65-CR-3
|
|
Address correspondence to: Shaun J Kilty Department of Otolaryngology – Head and Neck Surgery Room 242, 1053 Carling Avenue, Ottawa ON Canada. Phone: 1(613) 761-4030 Fax: 1(613) 729-2412 Email: shaunkilty@hotmail.com |
|
[HTML Abstract]
[PDF Full Text]
|
| How to cite this article: |
| Kilty SJ. Rapid diagnosis of sinonasal cancer in an anticoagulated patient: Outpatient clinic use of a gelatin-thrombin matrix (Floseal) for hemostasis following endonasal tumor biopsies. International Journal of Case Reports and Images 2011;2(11):10-13. |
|
Abstract
|
|
Introduction:
The purpose of this case report is to describe the use of gelatin-thrombin matrix (Floseal) for hemostasis to control severe nasal bleeding following endonasal biopsies in an elderly patient receiving anticoagulant therapies.
Case Report: An 81-year-old woman with advanced Alzheimer's disease presented with rapid onset of midfrontal swelling, pain and orbital hypertelorism. Profuse bleeding occurred following tumor biopsies in the outpatient clinic which was controlled in less than five minutes with a single administration of Floseal. The pathologic diagnosis was diffuse large B-cell lymphoma and treatment was initiated within the week. There was no recurrence of bleeding following this single treatment. The use of Floseal safely and economically allowed for rapid diagnosis and treatment of this sinonasal cancer without the need to stop and restart anticoagulation therapy in an elderly patient with dementia. Conclusion: The outcome from the clinical management of this case suggests that this treatment approach may be a model for use in the rapid diagnosis of sinonasal pathology in the outpatient clinic even in the setting of anticoagulation. | |
|
Key Words:
Floseal matrix, Hemostasis, Biopsy, Anticoagulants, Diagnosis, Warfarin, Aspirin, Paranasal sinuses
| |
|
Introduction
|
|
Floseal (FloSeal Hemostatic Matrix; Baxter Healthcare Corporation, Deerfield, IL, USA) comprises a gelatin matrix containing a reconstituted thrombin solution. [1] This topical hemostatic agent has been found to be useful for both problematic and severe bleeding encountered during surgery and other procedures at various anatomical sites. [1] Although the utility of this agent has been well described in the surgical setting, it may also have numerous clinical applications. This case report is the first to describe the use of Floseal in an anticoagulated elderly patient in an outpatient clinic, who required endonasal biopsy of a rapidly expanding sinonasal cancer to control the resultant bleeding. |
|
Case Report
|
|
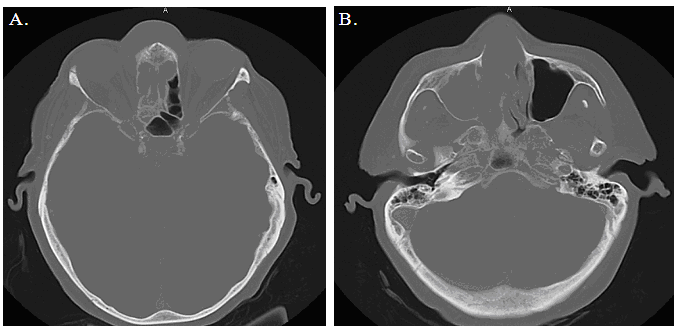
An 81-year-old woman was referred to the clinic for treatment of chronic rhinosinusitis (CRS). She had a history of recurrent pulmonary embolus and venous thrombosis from hereditary thrombophilia. Three years previously she had been diagnosed with Alzheimer's disease and dementia. She was a nonsmoker and otherwise healthy. Her medications included the anticoagulants warfarin and aspirin. The patient had experienced symptoms of CRS for about eight months. However, over the course of two weeks there was a rapid onset of midfrontal swelling and pain that was associated with orbital hypertelorism and a 15-pound weight loss. She did not have night sweats or fever. A physical examination revealed full extraocular movement without a pupillary defect. No deficits were found on cranial nerve examination. Endoscopy showed a mass filling the right nasal cavity, possibly emanating from the nasal septum. Externally, there was visible deformation of the midfrontal region. Recent bloodwork revealed sufficient anticoagulation (INR=2.6, PTT= 34 s). With informed consent from an accompanying family member, endonasal biopsies were performed under local anesthetic. As anticipated, this procedure was associated with profuse bleeding. A pre-prepared 5 ml intranasal instillation of Floseal was then applied to the biopsy sites. The nasal fossa was occluded for three minutes (the time for fibrin to polymerize per the manufacturer) and then irrigated with saline to remove any excess product that was not involved in clot formation. The bleeding was arrested in less than five minutes and there was no recurrence of bleeding following the Floseal treatment. Pathologically, a preliminary diagnosis of lymphoma was made and the patient underwent urgent imaging for staging (figure 1). Treatment for diffuse large B-cell lymphoma, the final pathologic diagnosis, with rituximab plus cyclophosphamide/vincristine/ prednisone (R/CVP) was then initiated within the week by a hematologist-oncologist. |
|
|
|
|
|
Discussion
|
|
Floseal has demonstrated efficacy as a hemostatic sealant to control perioperative and intraoperative bleeding in randomized clinical trials. [2] [3] [4] Primarily used as a hemostatic agent in vascular surgery, spinal surgery, and neurosurgery, Floseal has also been shown to provide safe and efficacious hemostasis during endoscopic sinus surgery and for the management of persistent or acute anterior epistaxis. [5] [6] [7] [8] Floseal has also been shown to decrease postoperative morbidity compared with electrocautery following adenotonsillectomy. [9] Additionally, the use of Floseal is associated with cost savings compared with electrocautery. [10] Notably, this gelatin-thrombin matrix has been shown to provide effective hemostasis in heparinized patients undergoing surgery. [2] Floseal is a biocompatible material combination of cross-linked bovine-derived gelatin granules and a reconstituted 500 IU/ml human thrombin component made from pooled human plasma. It is resorbed in 6-8 weeks as part of normal wound healing. Floseal is indicated in non-ophthalmic surgical procedures for the control of bleeding when ligature or other conventional treatment procedures are ineffective or not practical. Floseal works by activating the coagulation cascade by promoting contact activation of the intrinsic pathway and also by facilitating the conversion of fibrinogen to fibrin. Floseal is constituted clinically in four simple steps. First the thrombin solution is reconstituted from powder through the addition of 5 ml of calcium chloride. This solution is then transferred into a syringe containing the dry granules. The thrombin and granules are then mixed by passing the material between two syringes quickly for about 30 seconds to allow for adequate mixing. An applicator is attached to the syringe and Floseal is then applied to the bleeding site. To approximate the material at the site, it can be held with a gauze sponge or with similar technique. The bleeding may cease in as little as one minute with its maximum hemostatic effect occurring by 10 minutes. Once the bleeding is controlled, excess Floseal (Floseal not integrated into the clot) is then removed by irrigating with sterile saline. The precautions that must be taken with the use of Floseal include not directly applying it into blood vessels nor its use by injection into tissues. Similarly, it should not be used in individuals with known allergy to materials of bovine origin. Lastly, it should not be used for the closure of skin wounds. Floseal has several features that make it a particularly useful option for hemostasis in the outpatient clinic setting. First, the product is stored at room temperature and can be kept in treatment rooms for easy access. Second, the product can be quickly prepared for use (about two minutes). Third, the hemostatic effect is rapid; complete hemostasis can be achieved in a few minutes. Finally, Floseal is effective for any kind of bleeding when applied to the bleeding surface and it is therefore useful for many indications. This case illustrates the utility of Floseal in the setting of full anticoagulation in a patient with recurrent pulmonary embolus and venous thrombosis with hereditary thrombophilia. There was a particular need for rapid coagulation in this case, as the patient had advanced Alzheimer's disease and had difficulty following directives. The application of Floseal was easier, safer and provided a greater probability of success than attempting electrocauterisation to treat the profuse bleeding following biopsy in this vulnerable patient. The bleeding was fully treated within five minutes and there was no recurrence of bleeding. This diagnostic management plan using Floseal also prevented the potential morbidity or mortality from her stopping her regular anticoagulation therapies and avoided the repeated testing required for restarting anticoagulation therapy. |
|
Conclusion
|
|
This clinical case represents a model for an ideal clinical use of a topical hemostatic agent when tissue diagnosis is necessary in an anticoagulated patient. As in this case, its use allows for the rapid diagnosis and the subsequent initiation of treatment for lesions with a rapid evolution, such as diffuse large B-cell lymphoma, as was seen in this patient. Finally, the use of Floseal reduced the cost to the patient by reducing the time in the clinic and need for changes in her medications. The overall cost to the healthcare system was reduced by eliminating the need for repeated blood tests and physician/nurse visits that would have been required if she had stopped and re-started her anticoagulation therapies. Similarly, the large expense of an operating theatre and staff was avoided in this case. Overall, Floseal provided significant clinical and economic benefit in this case, and warrants both consideration and use in similar outpatient scenarios. |
|
Acknowledgement
|
|
Thanks to Julia R. Gage, PhD for assistance with writing the manuscript. |
|
References
|
|
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions:
Shaun J Kilty - Conception and design, acquisition of data and interpretation of data; Drafting the article and revising it critically for important intellectual content; Final approval of the version to be published |
|
Guarantor of submission:
The corresponding author is the guarantor of submission. |
|
Source of support:
None |
|
Conflict of interest:
Support for Dr. Gage and her assistance with this publication was provided by Baxter Healthcare Corporation. |
|
Copyright:
© Shaun J Kilty et. al. 2011; This article is distributed the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any means provided the original authors and original publisher are properly credited. (Please see Copyright Policy for more information.) |
|
|