|
 |
|
Case Report
| ||||||
| Primary thyroid diffuse large B cell lymphoma: A case report | ||||||
| Tej Prakash Soni1, Upendra Nandwana2, Lalit Mohan Sharma3, Anil Kumar Gupta4, Naresh Jakhotia1, Shantanu Sharma5 | ||||||
|
1Consultant, Department of Radiation Oncology,
Bhagwan Mahaveer Cancer Hospital and Research Centre,
Jaipur, Rajasthan, India 2Senior Resident, Department of Radiation Oncology, Bhagwan Mahaveer Cancer Hospital and Research Centre, Jaipur, Rajasthan, India 3Senior Consultant, Department of Medical Oncology, Bhagwan Mahaveer Cancer Hospital and Research Centre, Jaipur, Rajasthan, India 4Senior Consultant, Department of Surgical Oncology, Bhagwan Mahaveer Cancer Hospital and Research Centre, Jaipur, Rajasthan, India 5Associate Professor, Department of Radiotherapy, S.M.S. Medical College and Hospital, Jaipur, Rajasthan, India | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article] [Similar article in Pumed] [Similar article in Google Scholar] 
|
| How to cite this article |
| Soni TP, Nandwana U, Sharma LM, Gupta AK, Jakhotia N, Sharma S. Primary thyroid diffuse large B cell lymphoma: A case report. Int J Case Rep Images 2017;8(8):533–537. |
|
ABSTRACT
| ||||||
|
Introduction:
Primary thyroid lymphoma is a unique and rare entity. We report a rare case of primary thyroid lymphoma (diffuse large B cell lymphoma) in a male patient. Keywords: Differential diagnosis, Primary Thyroid lymphoma, Rare | ||||||
|
INTRODUCTION
| ||||||
|
Primary thyroid lymphoma is a rare entity. To diagnose it accurately and to differentiate with other thyroid malignancies is very important as primary thyroid lymphoma is highly curable without thyroidectomy. Most primary thyroid lymphomas are B cell non-Hodgkin’s lymphoma and usually treated by combination of chemotherapy and radiotherapy. | ||||||
|
CASE REPORT
| ||||||
|
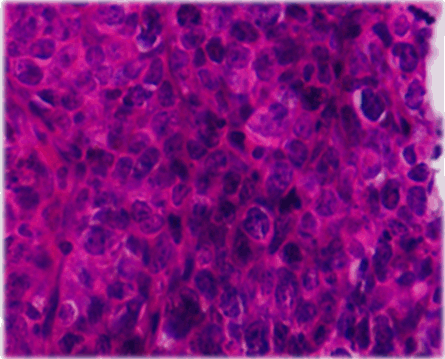
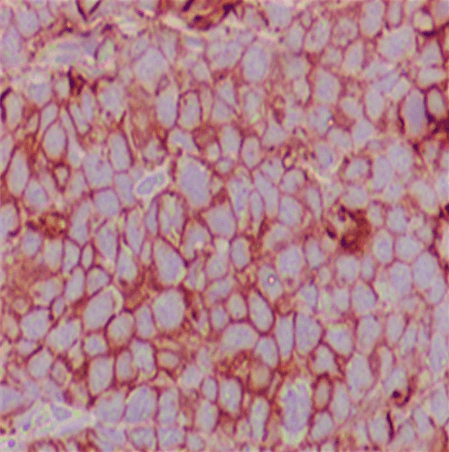
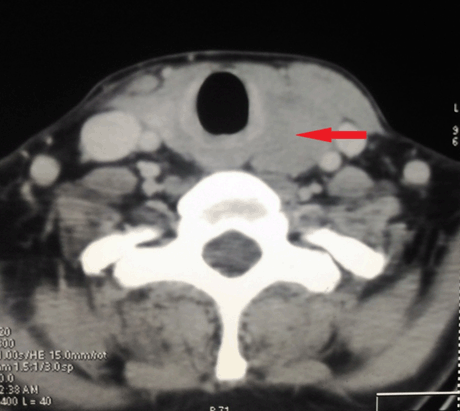
IA 40-year-old male with history of hypothyroidism, taking tab levothyroxine 100 mg once daily since one year presented to our hospital with the presenting complaint of a swelling in left side of neck, with progressive increase in size since last two months. On examination, a diffuse non tender lump measuring 6×3 cm in root of left side neck, moving with deglutition was noted with no palpable lymphadenopathy elsewhere. Serum T3, T4 and TSH levels were within normal limits. All other routine blood investigations were normal. Ultrasonography (USG) of neck showed mixed echogenic mass lesion in left lobe thyroid measuring 56x29 mm. Ultrasonography guided fine-needle aspiration cytology from the left lobe of thyroid lesion showed lymphoid large mononuclear cells. Trucut biopsy from the left thyroid lobe lesion was suggestive of non-Hodgkin’s lymphoma as lymphoid tissue with diffuse medium to large sized atypical cells with prominent round nucleoli and condensed chromatin with scanty cytoplasm and mitotic figures were present (Figure 1). Immunohistochemical analysis was reported as CD20 strongly positive, MIB-1 labeling index 60–65%, CD3 negative, CD5 negative, CD10 negative, CD23 negative, cycline D1 negative and Pan-Cytokeratin negative, suggestive of diffuse large B cell lymphoma (DLBCL) thyroid (Figure 2). The PET-CT scan showed FDG avid hypodense mass lesion involving the left lobe of thyroid gland measuring 63x38x31 mm with SUV Max 31.11 with no evidence of other abnormal lesion in the body (Figure 3). Bone marrow aspirate and biopsy were normal. Based on these investigations, final diagnosis was non-Hodgkin’s lymphoma-Thyroid (DLBCL, stage IEA). This case was discussed in multidisciplinary tumor board meeting. Chemotherapy followed by involved site radiotherapy (ISRT) as per our institute’s protocol was planned for him. He received four cycles of chemotherapy (retuximab, cyclophosphamide, doxorubicine, prednisolone: R-CHOP) under medical oncology unit. He tolerated the chemotherapy well. After chemotherapy there was complete remission of thyroid lesion. Involved site radiotherapy (ISRT) was given to the thyroid region to a dose 30 Gy in 15 fractions in three weeks. He is on regular follow-up. After one year of follow-up his disease is well controlled. | ||||||
| ||||||
| ||||||
| ||||||
|
DISCUSSION
| ||||||
|
Primary thyroid lymphoma is a rare tumor and makes less than 5% of thyroid malignancies [1]. It constitutes no more than 2.5% of all lymphomas [1]. Most thyroid lymphomas are of B cell origin non-Hodgkin’s lymphoma [2]. Diffuse large B cell lymphoma (DLBCL) constitutes 50–80% of the primary thyroid lymphoma and remaining 20-30% are extra nodal mucosa associated lymphoid tissue (MALT) lymphomas. Other rare subtypes include follicular lymphoma (12%), Hodgkin’s disease (7%) [2]. Primary thyroid lymphoma is more common in 60–70 years old age group females (Female:male ratio 3:1) [1]. The most common presenting sign and symptom includes recently growing painless mass in neck thyroid region. It may be associated with fever, sweating and weight loss (Classic B-type symptoms) up to 20% of patients [3]. Dysphagia, stridor, hoarseness of voice may be seen in 30% cases due to compression by the mass over adjacent structures [3]. About 10–30% cases may also have hypothyroidism at the time of presentation [3]. Ultrasongraphy (USG) guided fine-needle aspiration cytology (FNAC) of the thyroid lesion is a simple, cost effective, accurate and rapid to perform investigation test for thyroid lesions. Fine niddle aspiration biopsy added with flow cytometry and immunohistochemical studies may be more sensitive and specific. Trucut needle biopsy is recommended to confirm the FNAC results or if FNAC results are insufficient or inclusive. Ultrasonography of the thyroid is very useful to characterize the thyroid lesion such as size and shape of the lesion, micro or coarse calcification, solid or cystic appearance, texture of the gland, blood flow within the lesion, proximity to blood vessels etc. Computed tomography scan neck is done to confirm the Ultrasonography findings, assessment of local invasion, lymph node metastasis, and evaluation of oro-naso-laryngopharynx and esophagus. The PET scan is useful for staging, detection of distant metastasis or second primary, radiotherapy treatment planning and post treatment response evaluation. Bone marrow biopsy is also required for staging. Differential diagnosis of primary thyroid lymphoma includes Hashimoto’s thyroiditis (HT), secondary thyroid lymphoma, anaplastic thyroid carcinoma. Hashimoto’s thyroiditis (autoimmune chronic lymphocytic thyroiditis) is the most prevalent autoimmune thyroid disorder [4]. Hashimoto’s thyroiditis characteristic feature is lymphocytic infiltration of the thyroid gland followed by fibrous replacement of the thyroid parenchymal tissue. Hashimoto’s thyroiditis is a well known risk factor and associated with 40-fold increased risk of developing primary thyroid lymphoma when compared to the general population [4]. Hashimoto’s thyroiditis is associated with more than 90% of the primary thyroid lymphoma and due to this close association often both can be misdiagnosed for one another on FNAC [4]. Advanced and disseminated lymphoma can involve thyroid as secondary thyroid lymphoma. As with widespread disease burden, secondary lymphoma of the thyroid has poor treatment outcome compared to primary thyroid lymphoma [5]. It is very important to differentiate between anaplastic thyroid carcinoma and primary thyroid lymphoma. Anaplastic thyroid carcinoma is one of the most aggressive malignancies, with a poor prognosis and mean survival six months while primary thyroid lymphoma has excellent treatment outcomes. Primary thyroid lymphoma is treated mainly by chemotherapy with radiotherapy while surgical resection is needed for resectable anaplastic carcinoma thyroid. The presence of non-cohesive atypical large cells, irregular nuclear membrane, vesicular nuclei with prominent nucleoli and presence of lymphoglandular bodies in the background as cytology features, supported by CK and EMA negativity in IHC goes against the diagnosis of anaplastic carcinoma thyroid [6]. Primary thyroid MALT lymphoma has indolent course with superior clinical outcome (five-year survival more than 90%) compared to DLBCL of thyroid [7]. When MALT lymphoma (stage IE) is incidental diagnosis after thyroidectomy done for another reason there is no need of adjuvant chemotherapy or radiotherapy [7]. Thyroid lymphoma staging is done according to Ann-Arbor classification. Thyroidectomy is not recommended for the treatment of primary thyroid lymphoma [8]. Chemotherapy (CHOP: Cyclophosphamide, doxorubicin, vincristine, prednisone) followed by radiotherapy is the mainstay of the treatment for primary thyroid lymphoma [3][7]. Addition of rituximab for CD20 positive DLBCL in combination with CHOP chemotherapy increases disease-free survival and life expectancy [9]. Combination of chemotherapy and radiotherapy has shown superior results in compare to chemotherapy alone [10]. Doria et al. [10] reported the superior results with combination of chemotherapy and radiotherapy in a retrospective study of 211 patients of stage IE and IIE thyroid lymphoma with the relapse rates of 7.7%, 37.1% and 43% for chemoradiotherapy, radiotherapy and chemotherapy alone, respectively. Local recurrence rates were 2.6%, 12.6% and 23%, respectively in favor of combination therapy. The five-year life expectancy after the treatment of primary thyroid DLBCL is 75% with excellent treatment outcomes [7][10]. Stage III or IV, tumor size over 10 cm, lymph node involvement, mediastinal involvement, and presence of B symptoms are poor prognostic factors [7]. | ||||||
|
CONCLUSION
| ||||||
|
Primary thyroid lymphoma is a rare and unique disease, more commonly seen in females of 60–70 years age group. They present with rapidly growing masses in the thyroid gland region. Hashimoto’s thyroiditis is commonly associated with primary thyroid lymphoma. Ultrasonography guided fine-needle aspiration cytology and true cut needle biopsy from the thyroid lesion is required for the diagnosis and to differentiate with anaplastic carcinoma thyroid. Surgery/thyroidectomy is not recommended for primary thyroid lymphoma as chemotherapy and radiotherapy has excellent results and treatment outcomes. | ||||||
|
REFERENCES
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Tej Prakash Soni – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Upendra Nandwana – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Lalit Mohan Sharma – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Anil Kumar Gupta – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Naresh Jakhotia – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Shantanu Sharma – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2017 Tej Prakash Soni et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|