|
 |
|
Case Report
| ||||||
| Hyperleucocytic chronic myeloid leukemia with facial nerve palsy at presentation | ||||||
| Adama Isah Ladu1, Aisha Mohammed Abba2 | ||||||
|
1MBBS, MSc, FMCPath, Lecturer 1 and Consultant Hematologist, Department of Hematology and Blood Transfusion, University of Maiduguri Teaching Hospital, Borno State, Nigeria 2MBBS, FMCPath: Lecturer 1 and Consultant Hematologist, Department of Hematology and Blood Transfusion, University of Maiduguri Teaching Hospital, Borno State, Nigeria | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article] [Similar article in Pumed] [Similar article in Google Scholar] 
|
| How to cite this article |
| Ladu AI, Abba AM. Hyperleucocytic chronic myeloid leukemia with facial nerve palsy at presentation. Int J Case Rep Images 2017;8(8):523–527. |
|
ABSTRACT
| ||||||
|
Introduction:
Chronic myeloid leukemia (CML) is a clonal malignancy arising from the hemopoeitic stem cell (HSC). The disease can be complicated by hyperleucocytosis which is a medical emergency associated with high mortality of about 20–40%, and can results in various complication including tumour lysis syndrome, disseminated intravascular coagulation and leucostasis. The common sites for leucostasis are the lungs and central nervous system. Keywords: Chronic myeloid leukemia, Hyperleukocytosis, Facial nerve palsy | ||||||
|
INTRODUCTION
| ||||||
|
Chronic myeloid leukemia (CML) is a clonal malignancy arising from the hematopoietic stem cell. The hallmark of the disease is characterized by the presence of Philadelphia chromosome. This arises from the reciprocal translocation between the Abelson gene (ABL) on the long-arm of chromosome 9 and the breakpoint cluster region (BCR) on the long-arm of 22 [t(9,22) [1]. Hyperleukocytosis is defined as a white blood cell level of >100x109/L, and can complicate any form of leukemia. This is a medical emergency associated with high mortality of about 20–40%, and can results in various complication including tumor lysis syndrome, disseminated intravascular coagulation and leukostasis [2][3]. The common sites for leukostasis are the lungs and central nervous system (CNS). There are previous reports of adolescent patient with CML presenting with severe sensory impairment as a result of hyperleukocytosis complicated by CNS leukostasis [4]. Patients with overt CNS leukemia may manifest with cranial nerve palsy, commonly with unilateral seventh nerve palsy and less often the third and sixth cranial nerve [5]. In this report, we present a case of CML in an adolescent girl with hyperleukocytosis and seventh cranial nerve palsy at diagnosis. | ||||||
|
CASE REPORT
| ||||||
|
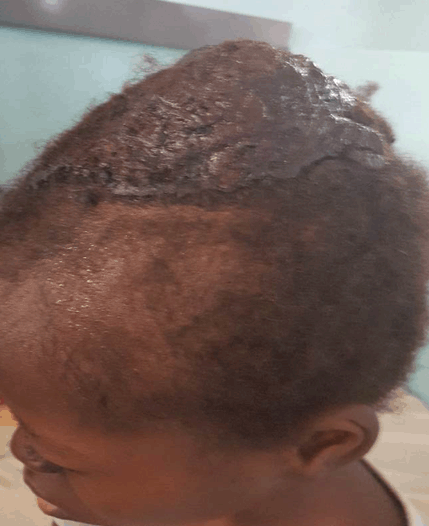
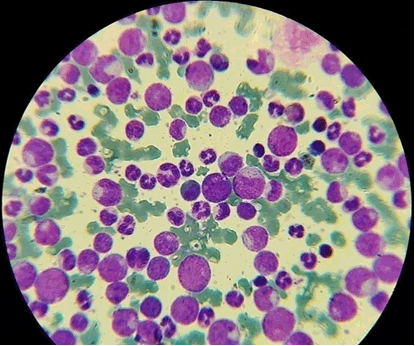
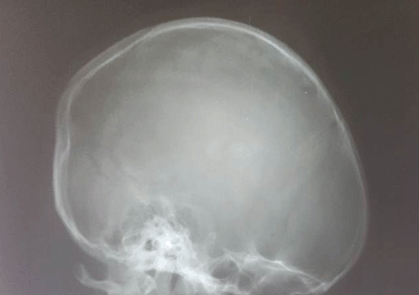
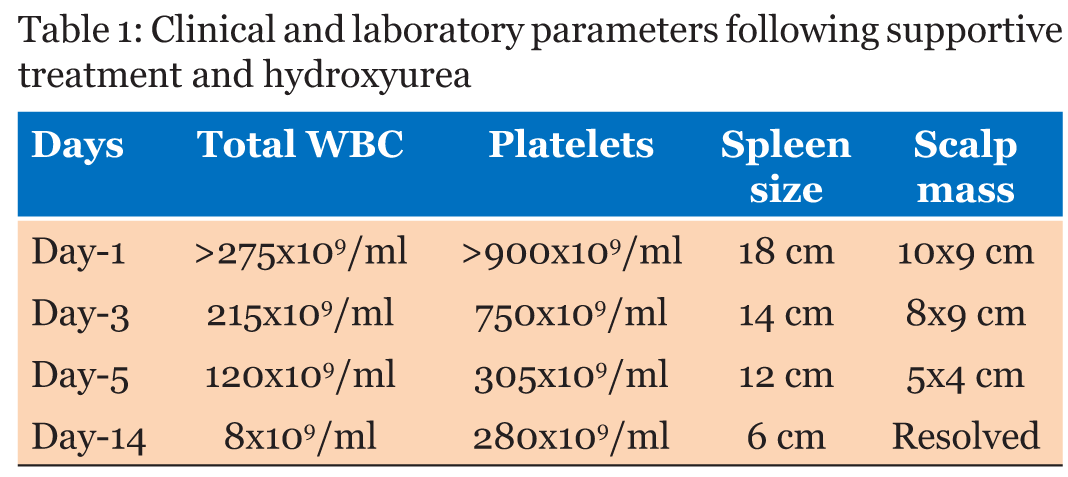
A previously healthy 15-year-old girl was admitted with one-month history of moderate to severe headache, weight loss, fever; one week prior to presentation she developed abnormal sensation in both ears, deviation of angle of the mouth to the left, with inability to close the right eye, and partial drooling of the left eye. Five days before admission, she developed a progressive scalp swelling which was initially the size of a pea, and said to have ruptured and bled spontaneously into the surrounding area, there was no other bleeding episodes. Her past medical history was unremarkable. General examination revealed an irritable and lethargic girl, sweaty, febrile (37.8°C), mildly pale, with a respiratory rate of 26 cycles per minute and pulse rate of 110 beats per minute, there was no peripheral lymphadenopathy. Local examination revealed no purpura or ecchymosis, however, there was a discreet, firm, moderately tender hemorrhagic mass on the scalp, measuring 10x10 cm (Figure 1). Neurological examination revealed no evidence of meningism; there was a right infra nuclear facial nerve palsy. There was partial ptosis of the left eye with otherwise normal ocular movement in all directions (Figure 2), both pupils were reactive to light and accommodation. No other focal neurology was evident. The spleen was palpable 16 cm and the liver 6 cm below their respective costal margins. Complete blood count revealed hematocrit of 28%, total white cell count of 275x109/L and platelets count of 980x109/L. Peripheral blood film (PBF) revealed all spectrum of white blood cells (Figure 3) –myeloblast 3%, promyelocytes 6%, myelocytes 40%, metamyelocytes 12%, band form 19% and neutrophil 20%. Bone marrow aspiration (BMA) cytology showed markedly increased myelopoiesis, the myelogram showed evidence of sequential maturation along the cell line with a peak at the myelocyte stage and myeloblast count of less than 5%. The PBF and BMA were in keeping with CML in chronic phase; rtPCR analysis of whole blood was positive for Philadelphia chromosome and the b3a2 transcript type (classical CML) was detected; quantification revealed BCR-ABL quantity of 4,578 copies per 2.5 micro liter of cDNA, ABL copies of 931,750 per 2.5 ml of cDNA and BCR-ABL/ABL ratio of 0.49%. The serum biochemistry was unremarkable. A skull X-ray revealed no bony involvement by the scalp swelling (Figure 4). Cerebrospinal fluid analysis was negative for malignant cells. Chest X-ray revealed cardiomegaly with right upper lobe opacity. Two-dimensional echocardiogram was performed and showed severe left ventricular dysfunction and dilated left ventricular wall, with increased pulmonary vascular resistance. An assessment of chronic myeloid leukemia presenting with hyperleukocytosis complicated by CNS leukostasis was made. Supportive treatment was started immediately with hydration using normal saline (150 ml/kg), allopurinol 100 mg daily and low dose aspirin (in view of the markedly raised platelets count) for 24 hr, before commencement of cytoreduction with hydroxyurea (40 mg/kg/day QDS). This was to expand the plasma volume and reduce the attendant risk of tumor lysis syndrome that may accompany cytoreduction. The patient showed progressive improvement in both clinical and laboratory parameters 72 hours after admission (Table 1). Her dose of hydroxyurea was titrated based on her total white blood cell count, and by two weeks following cytoreduction with hydroxyurea her WBC was stable at <10x109/L (Table 1). The scalp mass was managed conservatively with daily saline dressing, and resolved completely by day-10 of admission. The patient was referred to another tertiary centre in South west Nigeria where imatinib treatment made available by an international donor agency is given free of charge, however, her parents declined to proceed with any further treatment despite adequate counseling. She was continued on hydroxyurea and regular physiotherapy for her cranial palsy. She remained stable during regular follow-up, however, there was still residual seventh nerve palsy at third month follow-up. | ||||||
| ||||||
| ||||||
| ||||||
| ||||||
| ||||||
|
DISCUSSION
| ||||||
|
Neurological complications in patients with leukemia may result acutely either from the disease at the time of diagnosis or from relapse of the disease or as complication of treatment procedure [6][7]. Advance CNS involvement presents with features such as irritability, headache, seizures and coma. Overt CNS involvement is defined as the presence of white blood cell of five microliters or more in the CSF with evidence of blast cells and or presence of cranial nerve palsy or cerebral mass. This is seen in about 3% of children presenting with leukemia at the time of diagnosis [6]. The poor CNS perfusion following leukostasis or thrombosis in cases presenting with hyperleukocytosis may manifests with altered mental status. The high leukocrit associated with the raised total white blood cell count results in increased blood viscosity, consequently, the large non deformable blast cells results in reduce blood flow in marginally sized vessels and occlusion of microvasculature in the lungs and CNS [6]. The mainstay of management of symptomatic patients presenting with hyperleukocytosis consist of supportive treatment with aggressive hydration, allopurinol to prevent tumor lysis syndrome, urine alkalinization and appropriate respiratory support, followed by cytoreduction treatment using leukapheresis or chemotherapeutic agents like hydroxyurea [1][2]. In the index case, the patient presented with hyperleukocytosis, which was complicated by facial palsy. The facial nerve is the commonest involve in cranial nerve neuropathy [8][9]. There are few reports of children with AML presenting with facial nerve palsy at diagnosis [9][10]. However, it is rare manifestation in children with CML at the initial presentation. The time from the appearance of facial nerve palsy to diagnosis of leukemia varies from I day to I month; and usually improves one to six month following chemotherapy [10]. Allogenic stem cell transplantation with whole brain irradiation has been shown to be effective in facial nerve palsy in children with AML [10]. However, there is no definite therapeutic management in patients with CML, timely and prompt intervention in patients presenting with hyperleukocytosis can reverse some of the complications [6]. Of note in the index case is the cutaneous hemorrhage which may be related to the markedly raised platelets count, and the partial left eye drooling with an otherwise normal third nerve function; in addition, the patient had evidence of alveolar infiltrates on chest X-ray and other symptoms suggestive of hyperviscosity, which improved dramatically within few days of supportive treatment and hydroxyurea. | ||||||
|
CONCLUSION
| ||||||
|
In conclusion, presence of cranial nerve neuropathy at the initial presentation of patients with hyperleukocytosis indicates central nervous system involvement and requires prompt intervention to help reverse some of the features. Timely and aggressive intervention is particularly critical in poor resource setups like ours, where facility and expertise in leukapheresis is not readily available and therefore supportive management must be optimized. | ||||||
|
REFERENCES
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Adama Isah Ladu – Substantial contributions to concept and design of report, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Aisha Mohammed Abba – Substantial contributions to concept and design of report, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2017 Adama Isah Ladu et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|