|

|
 |
|
Case Report
| ||||||
| Coexistence of papillary carcinoma and anaplastic large cell lymphoma in a healthy 26-year-old male: A first case report | ||||||
| Majdi Saada1, Mechal Weiler2, Yaneev Zohar3, Ayelet Raz-Paster4 | ||||||
|
1MD, Resident at Internal a Department, Rambam Medical Campus, Haifa, Israel 2MD, senior doctor at Nuclear medicine department, Rambam Medical Campus, Haifa, Israel 3MD, senior doctor at the Pathology Department, Rambam medical Campus, Haifa, Israel 4MD, A, Head of Internal a Department, Rambam Medical Campus, Haifa, Israel | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] 
|
| How to cite this article |
| Saada M, Weiler M, Zohar Y, Raz-Paster A. Coexistence of papillary carcinoma and anaplastic large cell lymphoma in a healthy 26-year-old male: A first case report. Int J Case Rep Images 2017;8(4):265–269. |
|
Abstract
| ||||||
|
Introduction:We present an extremely rare presentation case of simultaneous existence of anaplastic large T cell lymphoma along with papillary thyroid carcinoma in a previously healthy 26-year-old male.
Keywords: Colon, Cyst, Duplication | ||||||
|
Introduction
| ||||||
|
Anaplastic large cell lymphoma (ALCL) is a subgroup of the peripheral T cell lymphomas (PTCL) which are a heterogeneous group of generally aggressive neoplasms that constitute less than 15% of all non-Hodgkin lymphomas in adults [1]. The ALCLs are further subdivided into three categories according to clinical criteria and immunohistochemistry: primary systemic anaplastic lymphoma kinase (ALK)(+), primary systemic ALK(-), and primary cutaneous ALCL [2]. Most patients with ALK negative ALCL are adults (age range 40–65 years) with a slight male predominance [3] . Thyroid cancer accounts for only 0.5–1.5% of all malignancies [4]. Papillary thyroid carcinoma (PTC) accounts for over 85% of all malignant thyroid tumors in regions with sufficient iodide intake. Risk factors for the development of thyroid cancer include radiation exposure, family history of thyroid cancer and hepatitis C related chronic hepatitis [5] [6] [7]. Herein, we report a clinical case of a 26-year-old-male with ALK(-) ALCL and papillary carcinoma of the thyroid which appeared after five months following chemotherapy treatment. | ||||||
|
Case Report
| ||||||
|
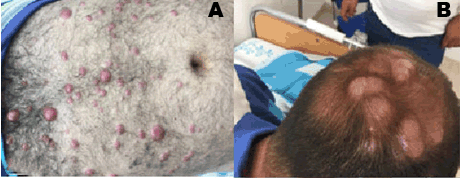
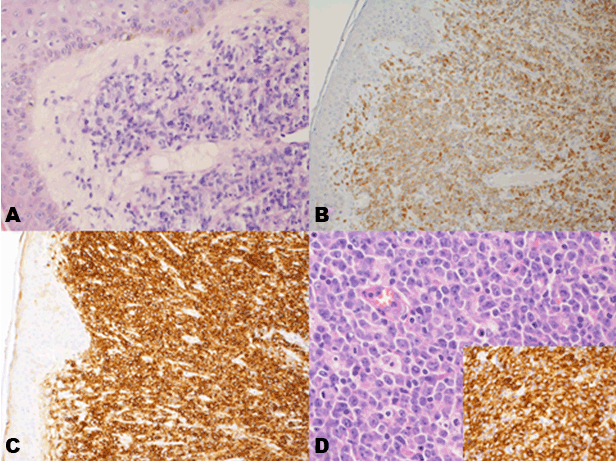
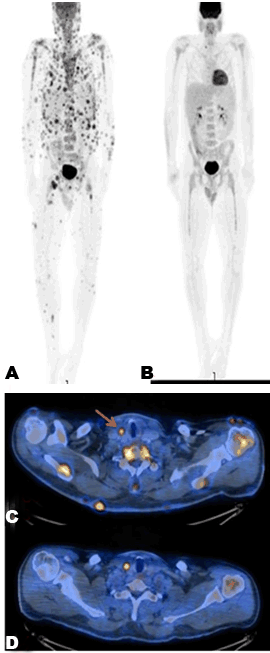
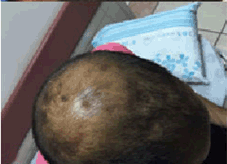
A previously healthy 26-year-old male presented with a history of fever, headache and subcutaneous nodules on the scalp of three weeks’ duration. The initial evaluation including physical examination, blood tests, brain CT and lumbar puncture was non-diagnostic. The patient’s skin lesions were suspected to be multiple scalp trichilemmal cysts and the patient was discharged to ambulatory follow-up. Two months later, he was admitted to the emergency department complaining of a neck pain, chest pain, dyspnea, general weakness and fatigue since two weeks. In addition, he reported a weight loss of six kilograms during a two-month period. On examination, the patient was fully oriented, his body temperature was 38.1°C, the heart rate was 105 beats per minute, the blood pressure was 130/70 mmHg and oxygen saturation was 96% while he was breathing ambient air. Skin examination revealed a firm nodular purple rash on the chest and the abdomen in addition to a soft nodule on the posterior scalp (Figure 1). A lymph node was enlarged on the left inguinal region. Lungs were clear on auscultation, there were no heart murmurs, and the abdomen was soft with no hepatosplenomegaly. Neurological examination revealed a supple neck with no other focal signs, the rest of the physical examination was otherwise normal. A complete blood count showed white blood cells of 10900 with 80% neutrophils, 18% lymphocytes and 0.6% eosinophils. The erythrocyte sedimentation rate was 70 mm/hr, the alkaline phosphatase level was 152 U/L, the lactate dehydrogenase level was 630 U/L, and serological tests for viral hepatitis B and hepatitis C were negative. A lumbar puncture was performed showing no evidence of central nervous system infection. Computed tomography scan of the chest showed no lymphadenopathy. Computed tomography scan of the abdomen showed enlarged liver and spleen, enlarged left inguinal lymphatic nodes 3–5 cm in diameter. Histopathologic examination from one skin nodule and from the inguinal node revealed infiltration by large T cell lymphocytes which were stained positively for CD3, CD4, CD30 and MUM1 and negative for CD8, TIA1, ALK, CLUSTERIN, CD123, CD56, CD10, CD20 and TDT (Figure 2). A proliferation index of 80% was reported. A bone marrow revealed hypercellular infiltration with T cells consistent with ALK negative ALCL. A 18F-fluorodeoxyglucose positron emission tomography computed tomography scan (FDG-PET-CT) showed nodal involvement including the nasopharynx and extranodal involvement including disseminated skin involvement and a lung lesion (Figure 3). A diagnosis of aggressive ALK negative ALCL stage IVB was made and treatment with CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone) and high dose methotrexate was begun. Following five cycles of the mentioned chemotherapy the patient felt well and there were notable improvement of the skin lesions (Figure 4). The patient underwent autologous hematopoietic stem transplantation with minor complications. A follow-up FDG-PET-CT performed five months after the initiation of treatment, showed resolution of all disease sites and pathological focal uptake at the upper right lobe of the thyroid gland (Figure 3). Ultrasound of the thyroid showed a solid echogenic nodule in the right lobe measuring 5.2x7.7 mm and another small nodule in the lobe of less than 5 mm. A fine needle aspiration showed findings consistent with papillary carcinoma of the thyroid. The patient was referred to a surgeon to undergo a total thyroidectomy. Histopathological examination from the thyroid confirmed the same diagnosis. On subsequent follow-up two months later; the patient reported feeling well with no traces of any of his diseases. | ||||||
| ||||||
|
| ||||||
|
| ||||||
| ||||||
|
Discussion
| ||||||
|
Papillary carcinomas of the thyroid are the most common malignant growth affecting the thyroid, currently representing 60–65% of malignant thyroid neoplasm. Most patients with papillary thyroid carcinomas present clinically with the discovery of a thyroid nodule during an unrelated physical exam or by the patient or others who notice a lump in the patient’s neck [8] [9]. Although the etiology of this neoplasm is unknown, they are thought to be related to neck irradiation, adenoma transformation, Hashimoto thyroiditis, family history of thyroid cancer, occupational and environmental exposures [9], None of which our vignette meets. The simultaneous existence of papillary carcinoma and other tumors had been reported in literature in rare cases, including the coexistence with squamous cell carcinoma in the thyroid and anaplastic carcinoma [10] [11] .The development of papillary carcinoma after chemotherapy is very rare, with one case report describing the development of PTC ten years after treating stage 1A Hodgkin’s lymphoma with ABVD (adriamycin, bleomycin, vinblastine, dacarbazine) chemotherapy [12]. Overall, papillary carcinomas represent an indolent group of neoplasm and have an excellent prognosis. Anaplastic large cell lymphoma is a peripheral T cell derived malignancy, representing around 2–3% of all lymphoid neoplasms. It is an aggressive lymphoma which frequently presents in an advanced clinical stage with systemic symptoms and extranodal involvement [13]. In a case of a systemic ALCL, pathological FDG uptake at the neck region can be seen as a part of the disease itself [14]. The coexistence of PTC with other lymphomas is rare, with 2 cases reporting the concurrent existence of thyroid MALT lymphoma and primary thyroid diffuse large cell lymphoma, respectively[15] [16]. Likewise, CD30+ T cell lymphoma and concurrent solid carcinoma tumor is a rare clinical scenario, with previous cases in literature reporting the co-existence with gastric, esophageal and renal carcinomas [17][18] [19]. This is the first clinical report of concurrent ALCL with PTC. The pathophysiology of the underlying mechanism is poorly understood, with some theories relating to monotypic expansion of activated T cells that occurs with solid tumors, and it is possible that T cell lymphoma originates from cytotoxic CD30+ T cell expansion [20]. Secondary genetic abnormalities probably lead to the dominance of a T cell clone, leading to irreversible transformation to lymphoma [21]. Whether ALCL predisposes to, or is merely associated with papillary carcinoma, is still a controversial issue which requires a more detailed consideration. | ||||||
|
Conclusion
| ||||||
|
Patients with anaplastic large cell lymphoma (ALCL) can frequently present with pathological uptake on PET-CT, thus easily masking an underlying solid tumor. Moreover, this case illustrates the need for further research on the underlying mechanism of the seemingly related tumors. | ||||||
|
Acknowledgements
| ||||||
|
We are thankful to Ali Yahya, Alaa Khateeb and Noa Lavi for their contribution to the study. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Majdi Saada – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Mechal Weiler – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Yaneev Zohar – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Ayelet Raz-Paster – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2017 Majdi Saada et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|