|
 |
|
Case Report
| ||||||
| Walking the thin line between malignancy and infectious disease: A case report | ||||||
| Catarina Couto1, Sofia Fraga1 | ||||||
|
1MD, Pediatrics’ Resident, Hospital Garcia de Orta, Almada, Portugal
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article] [Similar article in Pumed] [Similar article in Google Scholar] 
|
| How to cite this article |
| Couto C, Fraga S. Walking the thin line between malignancy and infectious disease: A case report. Int J Case Rep Images 2017;8(12):762–769. |
|
ABSTRACT
| ||||||
|
Introduction: Children infected by human immunodeficiency virus have a higher incidence of malignancy than non-infected children. Keywords: Diffuse large B cell lymphoma, Human immunodeficiency virus, Malignancy | ||||||
|
INTRODUCTION
| ||||||
|
Late diagnosis of human immunodeficiency virus (HIV) infection in children can be associated with a more severe presentation because HIV-related immunosuppression increases the incidence of an acquired immune deficiency syndrome (AIDS). HIV-infected children have a higher incidence of malignancy than non-infected children, especially if they are not receiving treatment with highly active antiretroviral therapy [1][2][3]. AIDS malignancies include Kaposi’s sarcoma, non-Hodgkin lymphoma and invasive cervical cancer. | ||||||
|
CASE REPORT
| ||||||
|
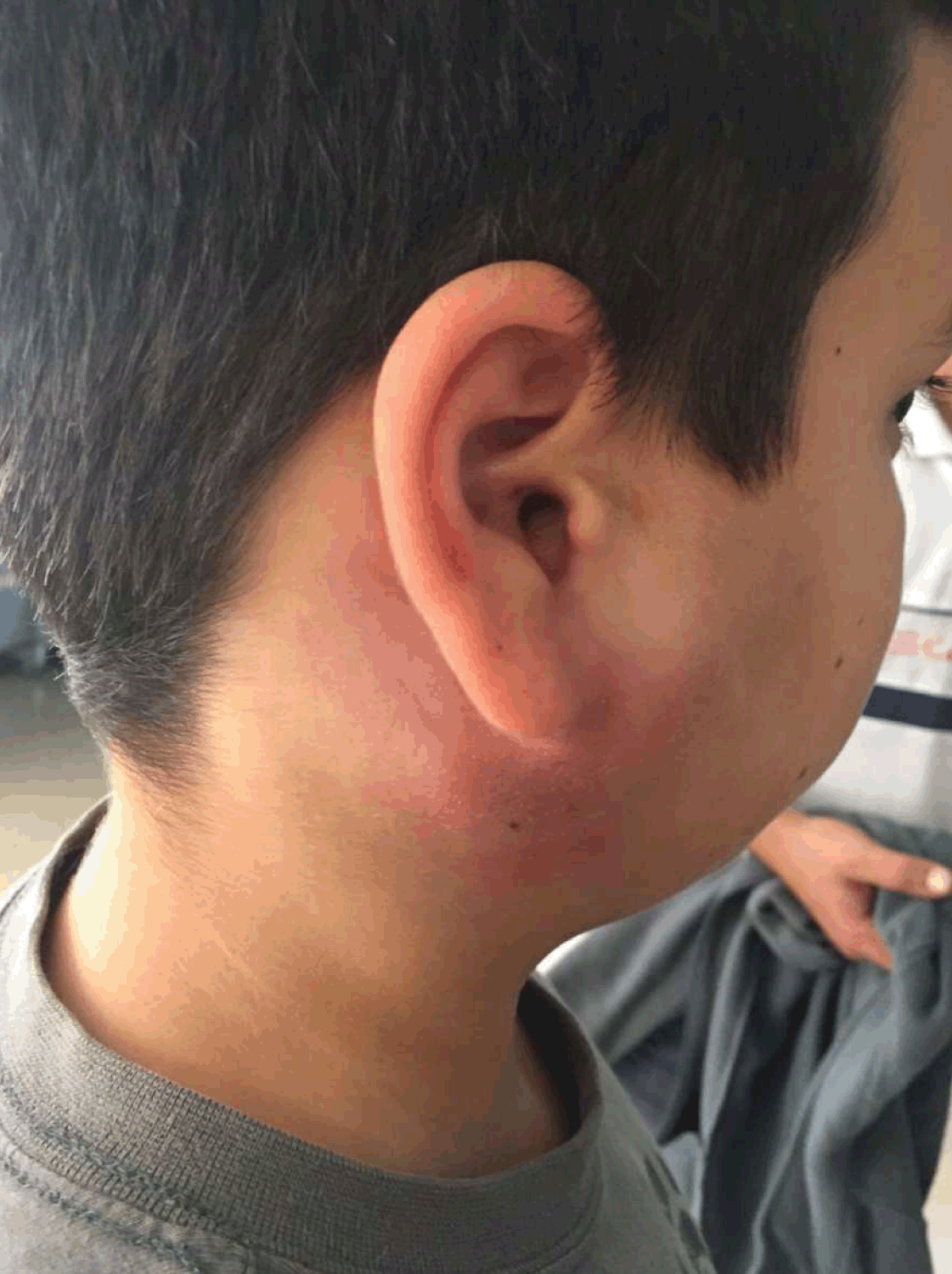
An apparently healthy 13-year-old boy presented to the A&E department of his local hospital with a month long history of a painless swelling of the right parotid gland which extended to the right cervical region, with no signs of skin infection, fever, weight loss or any other constitutional symptoms. The patient denied any current medication, animal exposure, recent traveling or ingestion of unpasteurized dairy products or undercooked meats. His personal and family history, as were given at the time, were uneventful. An ultrasound was performed which showed a nodular structure within the parotid gland of approximately 2.4 cm in diameter, described as a probable adenopathy. The ultrasound also identified multiple small right laterocervical lymphadenopathies. To better ascertain the etiology of the mass, a magnetic resonance imaging (MRI) scan revealed an increase in the size of the parotid gland and a nodular lesion within the gland with central areas of necrosis. He was started on oral cefradine, a first generation cephalosporin, which he continued for 15 days with no signs of improvement. At this time he was referred to a pediatric oncology center to exclude a possible lymphoma. On admission to the center, he had a fine-needle aspiration cytology and flow cytometry which were both negative for neoplastic cells. He also had an extensive blood workup performed (Table 1) which showed a thrombocytopenia, a slightly increased erythrocyte sedimentation rate and lactate dehydrogenase and a hypergammaglobulinemia. Multiple viral serologies were also performed, including HIV p24 antigen, which was positive. A confirmatory test for human immunodeficiency virus (HIV) infection was performed and was also positive. We then learned that both parents had been diagnosed with HIV infection when the child was four-year-old and no testing had been done on the boy at the time. After HIV diagnosis, the child was referred to our hospital where he would have HIV follow-up. On admission, the patient was a healthy looking boy with a body mass index of 21.3 kg/m2, he had a swelling of the right parotid gland and cervical region with a slight erythema (Figure 1), palpable right cervical lymphadenopathies and no hepatosplenomegaly. The rest of the examination was normal. His initial viral load was 96,730 cp/mL and his CD4 count was 189 cells/mL (Table 2). He was tested for antiretroviral resistance, which was negative, and had a tuberculin skin test, which was negative. He was started on Atripla® (emtricitabine+tenofovir+efavirenz) and cotrimoxazole. He was also started on amoxicillin and clavulanic acid, which he maintained for 15 days, because he had some signs of skin infection. After three weeks of antibiotics, the swelling and erythema of the right parotid gland continued to evolve (Figure 2) and he was switched to clindamycin for another 15 days with no response (Figure 3). At this point tuberculous lymphadenitis was suspected, the patient had a chest X-ray, which was normal, and a cervical computed tomography (CT) scan which showed a right cervical conglomerate of lymphadenopathies with areas of necrotic tissue, multiple small left cervical and bilateral supraclavicular lymphadenopathies (Figure 4). A lymph node aspiration was performed but came back as having insufficient material so the patient had a lymph node biopsy. Due to the clinical suspicion of tuberculous lymphadenitis and despite having a negative acid-fast bacilli stain, he was started on isoniazid, rifampicin, pyrazinamide and ethambutol but there was no improvement (Figure 5). We then learned the biopsy revealed a diffuse large B-cell lymphoma. He was started on chemotherapy. The culture for Mycobacterium tuberculosis was negative and he stopped the tuberculous drugs. The final diagnosis was diffuse large B cell lymphoma, an AIDS-defining disease in a perinatally HIV-infected patient. The patient completed three months of chemotherapy, without any major complications, and at sixth month follow-up postchemotherapy, he is in remission. He is still on the same HAART regimen and now has an undetectable viral load. Immune reconstitution has been slow and his CD4 count is still under 200 cells/µL (Table 2). | ||||||
|
||||||
| ||||||
|
||||||
| ||||||
| ||||||
|
| ||||||
| ||||||
|
DISCUSSION
| ||||||
|
Mother-to-child transmission (MTCT) is the principal mode of HIV infection in children (92% of HIV-infected children in Portugal have acquired it perinatally) [4]. Since early 2004, nationwide HIV screening during pregnancy (1st and 3rd trimester) has been mandatory in Portugal. Regularly testing for HIV infection in pregnant women is one of the most important preventive measures of MTCT of HIV because it allows for early treatment with highly active antiretroviral therapy (HAART), which will decrease the viral load and improve immune function and thus reduce the risk of transmission to the fetus and newborn. In Portugal, around 30% of HIV-infected mothers are diagnosed during pregnancy [4]. Despite such a high rate of maternal diagnosis during pregnancy, which is probably explained by the considerable number of women from foreign countries who come to Portugal during pregnancy (44% of HIV-infected mothers are of non-Portuguese nationality) [4], transmission rate has been consistently under 2% since 2005, which highlights the importance of testing during pregnancy. Some cases of mother-to-child transmission will keep evading early detection, like in the case reported. There are two reasons for late diagnosis of perinatally acquired HIV infection in a child: either the mother is in the window period in the 3rd trimester and, despite being infected, tests negative for HIV or the maternal infection occurs postnatally and the child is infected through breastfeeding. Perinatally infected infants usually progress earlier than older children or adults, because they are infected before complete maturation of the immune system. Newell et al. reported a mortality rate of 35.2% of HIV-infected infants in the first year of life [5]. In the CHER trial, 16% of untreated and 4% of treated HIV-infected infants died during the median follow-up of 40 months [6]. These infants are called rapid progressors. Those who present in childhood or adolescence are called slow progressors. The boy in the case reported is a slow progressor. He had no symptoms suggestive of HIV infection until the lymphoma developed. Using the Centers for Disease Control and Prevention classification system, he has a stage 3 HIV infection (CD4 count of less than 200 cells/mm3 and an AIDS-defining illness, non-Hodgkin lymphoma). Since the INSIGHT and TEMPRANO studies in 2015 [7][8], the paradigm for treatment guidelines regarding initiation of HAART in HIV-infected patients has changed and now HAART is recommended for all patients regardless of CD4 counts. Both these studies showed that early HAART was associated with a decline in severe morbidity. Early treatment was already recommended in infants under 12 months of age after the CHER trial proved a reduction of infant mortality by 76% and of HIV progression by 75% in infants who were immediately started on HAART versus those who were deferred until clinical criteria or CD4 percentages were met [6]. Despite there not being randomized controlled trials supporting early versus deferred treatment in children older than one year of age, PENTA has acknowledged that there is evidence of the long-term benefits of early HAART and since 2016 has recommended treatment for all children with HIV infection [9]. Once-daily antiretroviral regimens are available for HIV-infected older children and adolescents. Despite a theoretical advantage compared to twice-daily regimens in regards to better adherence, this was not proven in the Madrid children and adolescents cohort study [10]. Nevertheless simplification of HAART regimens should remain one of the goals of HAART prescription. In this case, we chose Atripla® (emtricitabine+tenofovir+efavirenz) because it is an extensively researched drug with a reasonable safety profile. The differential diagnosis of chronic localized lymphadenitis in HIV-infected children includes tuberculous lymphadenitis (when in the cervical region, this is called scrofula), non-tuberculous mycobacterial infection, cat scratch disease, HIV infection and lymphoproliferative disorders. A lymphoproliferative disorder was wrongly excluded earlier because this patient had had a fine-needle aspiration cytology and flow cytometry which were both negative for cancer cells. Since he had no history of contact with tuberculosis, we initially considered the HIV as the causative agent, associated with a bacterial skin infection. Considering there was no improvement with two courses of antibiotics and because the cervical CT findings of a large conglomerate of cervical lymphadenopathies with central areas of necrosis as well as supraclavicular adenopathies were suggestive, tuberculous lymphadenitis was considered as a possible diagnosis. Testing for active tuberculosis in HIV patients is made difficult by the unreliableness of both the tuberculin skin test and the interferon-gamma release assays in HIV-infected patients [11][12][13]. Culture of Mycobacterium tuberculosis is still the gold standard for diagnosis of tuberculosis infection, but the long incubation period of six to eight weeks makes it impractical. Though molecular techniques allow for a rapid diagnosis, in this case this method was not available. Screening for Mycobacterium tuberculosis infection was done on admission to the HIV clinic with a tuberculin skin test, which was negative, and a chest X-ray, which was normal. Both the acid-fast bacilli staining and culture of Mycobacterium tuberculosis were negative and the biopsy eventually revealed a non-Hodgkin lymphoma. HIV-infected children are at a higher risk of developing cancer than their non-infected counterparts. In literature, this ratio has varied from 10.08 (95% CI 5.87–16.14) in the Pediatric AIDS Clinical Trials Group [1] up to 2288 (95% CI 920–4715) in the report by Evans et al. [2]. The overall prevalence of malignancy in HIV-infected children has been reported as 3.6% in the pre-HAART era [2]. Highly active antiretroviral therapy (HAART) has reduced the incidence of Kaposi’s sarcoma and non-Hodgkin lymphoma, both AIDS-defining conditions [14][15]. This is related to the fact that HAART restores immune function and therefore, reduces incidence of all AIDS-defining conditions. However, non-AIDS-defining cancers have been on the rise in adults [16][17], mainly because of the growth and aging of the HIV population. There is some evidence that the rate of non-AIDS-defining cancers may be increasing in the pediatric population as well [15][18]. Potential explanations for the increased cancer incidence in HIV patients include HIV-related immunodeficiency, increased prevalence of known cancer risk (such as smoking), a direct pro-oncogenic effect of HIV through viral proteins such as tat and VPr, co-infections with known oncogenic viruses (Burkitt lymphoma is associated with the Epstein–Barr virus, Kaposi’s sarcoma with the HHV-8 and cervical cancer with the human papilloma virus) and the activated inflammatory and coagulation pathways [19][20]. Whereas in Europe and United States of America, the most common cancer in HIV-infected children is non-Hodgkin lymphoma [2], in Sub-Saharan Africa Kaposi’s sarcoma, which is associated with human herpes virus 8 (HHV-8), is more prominent [20]. Other cancers that are frequently associated with HIV infection include leiomyosarcoma and Hodgkin’s lymphoma [7][20]. HIV-infected children often present with more advanced malignancies than non-infected children [21]. This may be due to a faster proliferation of cancer cells unchecked by a deficient immune system [20][22]. As it happened in this case, a delay in cancer diagnosis is common, because clinical signs of HIV are often similar to those of cancer [20]. Problems in the management of cancer in HIV-infected children include a higher incidence of opportunistic and/or aggressive infections, possible drug interactions between HAART and cytotoxic drugs and multiorgan dysfunction caused by the virus [23]. Several studies have supported the initiation or maintenance of HAART during treatment with cytotoxic drugs, as HAART has been showed to increase the chance of remission and survival rates [3][18][23] though some concerns remain regarding potential drug interactions. Protease inhibitors and non-nucleoside reverse transcriptase inhibitors are known inhibitors or inducers of the cytochrome P450 enzymes [24] [25]. Through induction antiretroviral drugs can decrease the efficacy of cytotoxic drugs and through inhibition they increase their toxicity [24][25]. Despite this, there are no current guidelines on dose adjustment of HAART or chemotherapy drugs [24][26]. However, it seems reasonable to substitute zidovudine for another antiretroviral drug because of the added risk of bone marrow suppression. In the pre-HAART era, the prognosis of malignancy in HIV-infected children was very poor. Out of the seven HIV-infected children with non-Hodgkin lymphoma reported by Evans et al. in 1997, only one had survived at four years of follow-up [2]. This child was one of the only two who had had standard intensive chemotherapy. The median survival time for the remaining children was 6.5 months (2–14 months). Godot et al. [27] reported on the prognosis of B cell lymphomas in HIV-infected children in the HAART era with much better results. Out of 12 children with high grade B cell lymphoma, 8 had survived at a median follow-up of 72 months. Morbidity and treatment related toxicity were similar to what is reported for non-infected children. Studies in adults have suggested that the response rate to chemotherapy and overall survival in HIV-infected patients with non-Hodgkin lymphoma are approaching those seen in the general population when controlled for the same stage of disease [28][29]. | ||||||
|
CONCLUSION
| ||||||
|
Even though preventive measures of mother-to-child transmission of human immunodeficiency virus (HIV) have greatly reduced the incidence of pediatric HIV infection, some infants will continue to escape early detection because of late maternal diagnosis. The diagnosis of malignancy in HIV-infected patients is often made difficult by overlapping symptoms and a delayed diagnosis is common. | ||||||
|
REFERENCES
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Catarina Barreiros Couto – Substantial contributions to conception and design, Acquisition of data, Drafting of the article, Final approval of the version to be published Sofia Fraga – Acquisition of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of Submission
The corresponding author is the guarantor of submission. |
|
Source of Support
None |
|
Conflict of Interest
Authors declare no conflict of interest. |
|
Copyright
© 2017 Catarina Barreiros Couto et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|