|
 |
|
Case Report
| ||||||
| Esophageal dysmotility and abscess formation post Roux-en-Y bariatric surgery: A case report | ||||||
| Ryan D. Nicklas1, Daniel S. Swink1, Alyssa E. Heinrich1, Christopher Boyd2, Christina Li3 | ||||||
|
1MS, Trinity School of Medicine, Kingstown, SVG
2MD, MIS Fellow, Department of Bariatric and Minimally Invasive Surgery, Northwest Hospital Baltimore, MD, USA 3MD, FACS, Division Head, Department of Bariatric and Minimally Invasive Surgery, Northwest Hospital Baltimore, MD, USA | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article] [Similar article in Pumed] [Similar article in Google Scholar] 
|
| How to cite this article |
| Nicklas RD, Swink DS, Heinrich AE, Boyd C, Li C. Esophageal dysmotility and abscess formation post Roux-en-Y bariatric surgery: A case report. Int J Case Rep Images 2017;8(11):746–751. |
|
ABSTRACT
| ||||||
|
Introduction: Abscess formation in the postoperative bariatric patient can be the result of anastomotic leak, a dreaded complication of Roux-en-Y gastric bypass. However, there is little data on bowel obstruction disrupting the anastomotic site resulting in leak and esophageal dysmotility. Keywords: Abscess, Esophageal dysmotility, Roux-en-Y | ||||||
|
INTRODUCTION
| ||||||
|
Roux-en-Y is a safe and effective means of weight control in patient’s population who understand the potential risks, as well as the behavioral modifications required postoperatively. The procedure involves dividing the stomach and the jejunum and the rejoining the segments in a manner that bypasses a segment of the small bowel. By this process the absorptive length of the small bowel decreases and the volume of the remaining gastric pouch is restricted (Figure 1). There is convincing evidence that bariatric surgery can mitigate or even cure obesity related disease such as gastroesophageal reflux, sleep apnea, and type-2 diabetes. Furthermore, long-term mortality rates are lower for morbidly-obese patients who have undergone bariatric surgery than those who have not [1][2]. However, it is important to be aware of the potential postoperative complications and management in these patient populations. The most frequently described complications include leak, obstruction, and anastomotic ulcer [2]. Another complication is abscess formation, which can result from seeding of intestinal contents into the abdomen during surgery, or spillage from the anastomosis. Uncommonly, a bowel obstruction can lead to transient increase in intraluminal pressure leading to disruption of the anastomotic site. Both abscess and anastomotic leak require surgical intervention for complete resolution [2]. In this case, we aim to bring attention to the treatment and management of complex postoperative bariatric patients with obstruction, leak, dysmotility and consequential abscess formation. | ||||||
| ||||||
|
CASE REPORT
| ||||||
|
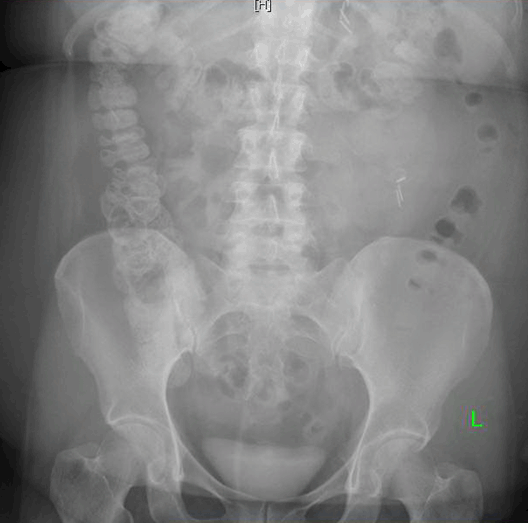
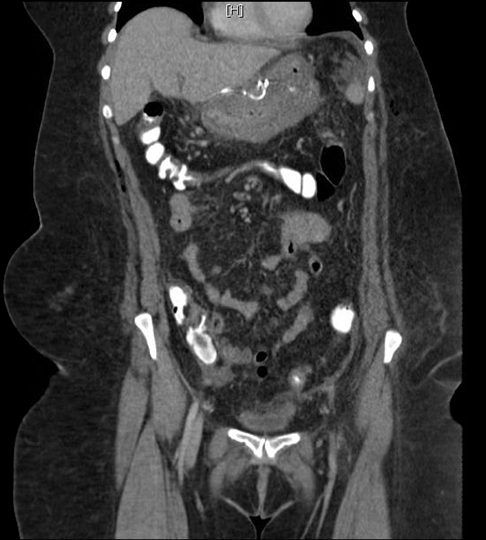
A 50-year-old female presented with a past medical history of gastroesophageal reflux, hypertension, hypothyroidism, inflammatory polyarthropathy, and obesity with a BMI of 44 kg/m2. Her surgical history included tonsillectomy, hysterectomy, cholecystectomy, and left knee replacement. Her medications included omeprazole, plaquenil, levothyroxine, and liothyronine. The patient underwent laparoscopic Roux-en-Y gastric bypass and hiatal hernia repair. She tolerated the procedure well, was functioning within normal limits, and was subsequently discharged on postoperative day-1. The patient was doing well on her follow-up appointment on postoperative day-5 where she was tolerating 80 oz of fluid a day; well above the minimum requirement. However, a few hours later the patient developed severe cramping epigastric pain, nausea, vomiting, and presented to the emergency department. The patient had a CT scan of her abdomen and pelvis, which showed obstruction at the level of jejunojejunostomy. There was no evidence of a leak or abscess (Figure 2). After intravenous fluid resuscitation, an X-ray of the abdomen and pelvis was performed eight hours later and showed transit of contrast into the ascending and transverse colon (Figure 3). However, given the lack of resolution of her symptoms, she was taken to the operating room urgently. A diagnostic laparoscopy was performed revealing a single adhesion of her jejunojejunostomy down to the base of the mesentery, effectively torsing the distal Roux limb. An adhesiolysis was performed. The patient recovered well, tolerated fluids, and was discharged one day later. However, at her follow-up appointment 12 days post Roux-en-Y and six days post adhesiolysis, the patient again developed nausea, vomiting, epigastric pain and dysphagia. She denied fevers, was not tachycardic, and had no signs or symptoms suggestive of infection. An outpatient Gastrografin swallow test was performed. The films were interpreted as showing no evidence of anastomotic obstruction or leak. However, it was suggestive of significant narrowing of the gastrojejunal anastomosis site and marked distal esophageal dysmotility as there was a five-minute delay of transit from the distal esophagus into the gastric pouch (Figure 4). Due to her persistent symptoms and impaired oral intake, the patient was directly admitted to the hospital. On admission, the patient received fluid resuscitation for dehydration and pharmacologic management of her nausea and vomiting with rectal Phenergan. The patient was afebrile, had a CBC and CMP within normal limits, and showed no other signs of infection on examination. Upper endoscopy was performed to rule out recurrence of hiatal hernia, stenosis from the repair, or anastomotic problems. It revealed no evidence of stricture or obstruction. Despite these findings, the patient’s persistent nausea, epigastric pain and clinical examination conflicted with these results. A diagnostic laparoscopy for possible revision of gastrojejunal anastomosis, evaluation of hiatal hernia repair, and possible endoscopy were planned for the next day. During the laparoscopy, the jejunojejunostomy did not appear obstructed, although a large amount of edema was appreciated around the distal gastric pouch and by the liver. While defining the borders of the gastrojejunal anastomosis and gastric pouch, there was a sudden rush of purulent fluid. A cavity of purulent fluid was revealed, extending below the left lobe of the liver and medial to the gastric pouch beneath the gastrohepatic ligament. The location of the abscess suggested possible impingement on the gastric pouch and pressure on the gastroesophageal junction. The purulence was completely aspirated, a Blake drain was placed in the abscess cavity, and a gastrostomy tube was placed in the gastric remnant for nutritional support. Intraoperative anastomotic leak test revealed no evidence of current leakage of contents into the abdomen. The remainder of the surgery was uneventful and the patient was placed on empiric piperacillin/tazobactam and infectious disease was consulted. Culture of the abscess revealed Hemophilus parainfluenza and Prevotella species, normal oral flora. Infectious disease recommended modifying antibiotic therapy to a two-week course of ampicillin/sulbactam and a repeat abdominal CT scan to evaluate for remaining abscesses. Computed tomography scan of abdomen and pelvis with intravenous contrast taken on day-1 post laparoscopy showed an edematous, postoperative appearance of the stomach, with gastric tube and drain in good positioning, and no evidence of remaining abscess (Figure 5). Clinically, the patient reported improvement in her abdominal pain and nausea. The patient was advised to continue to meet fluid and protein requirements, with a combination of PO and G-tube feeds, and to advance diet to soft foods as tolerated. One week after discharge, the patient was seen in the office and was doing reasonably well, she reported relatively controlled gastroesophageal reflux and nausea with zantac and zofran, respectively. By this visit the patient had lost 30% of her excess body weight. | ||||||
|
| ||||||
|
| ||||||
|
| ||||||
|
| ||||||
|
DISCUSSION
| ||||||
|
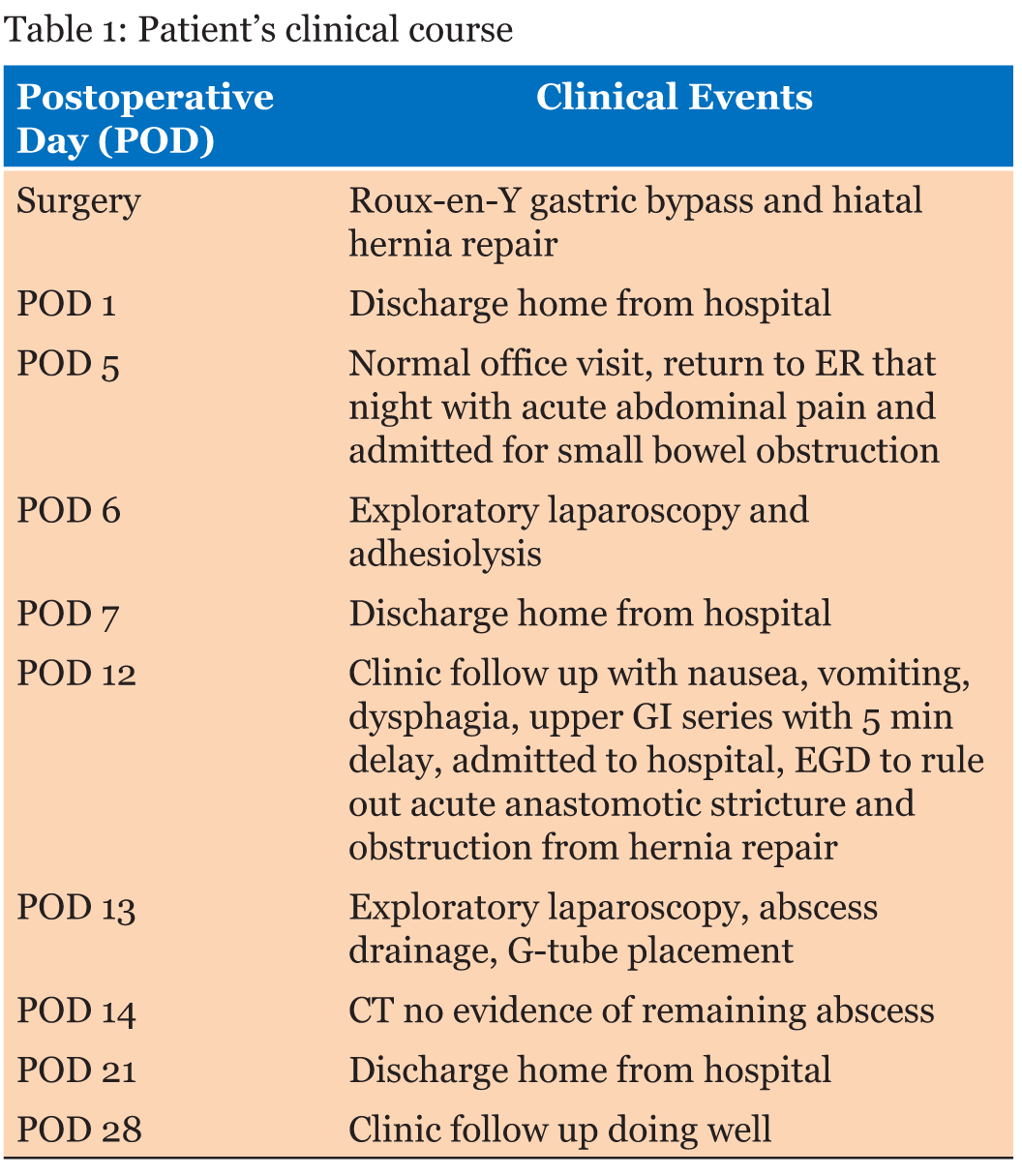
The American Society for Metabolic and Bariatric Surgery estimates that 23% of bariatric surgeries performed in 2015 were Roux-en-Y gastric bypass, making it the second most common bariatric procedure performed in the United States following gastric sleeve which comprised 54% of bariatric procedures [3]. Of Roux-en-Y procedures, less than 7% of laparoscopic cases are associated with major 30-day complications [4]. Exceptional in our patient, is the rapid onset of bowel obstruction, leading to progressive increase in intraluminal pressure, causing presumed disruption of the gastrojejunal anastomosis and leak, ultimately resulting in abscess formation and esophageal dysmotility by mass effect (Table 1). The patient’s initial postoperative hospitalization was for small bowel obstruction. Small bowel obstruction is a potential post Roux-en-Y gastric bypass complication. Small bowel obstruction occurs in 1–11% of patients postoperatively [5][6]. The timing of obstruction is highly variable, but one study suggests an average onset of 7–19 days postoperatively for jejunojejunostomy obstructions [7]. Another study suggests a mean time to presentation of 313 days after Roux-en-Y bypass [8]. The most common cause of obstruction, occurring in 42–54% of cases is due to internal hernias [2][7][8]. These are caused by defects in the mesentery created during the operation. Other sources of obstruction include mesocolon scarring as well as adhesions. In our patient, the obstruction was the result of an adhesion located at the jejunojejunostomy occurring only five days postoperatively. Laparoscopic surgeries generally carry a lower risk of intra-abdominal adhesions compared to open surgeries due to less tissue damage, and less organ manipulation [2][7]. While not specific; nausea, vomiting, and abdominal pain out of proportion to exam in the postoperative picture should lead to increased clinical suspicion for obstruction. Patients may be diagnosed with a CT scan and the treatment is early reoperation to prevent further complications [2][7]. Our patient’s obstruction was relieved by performing adhesiolysis shortly after her presentation to the emergency department, with rapid resolution of her symptoms. However, it would later be discovered that the obstruction contributed to additional complications later in her clinical course. The patient’s return with nausea, vomiting, and impaired oral intake, prompted evaluation with a Gastrografin swallow study, which identified evidence of alterations in normal esophageal function. Esophageal dysmotility has multiple etiologies and is traditionally not associated with Roux-en-Y gastric bypass surgery. In fact, Roux-en-Y is the preferred bariatric surgery in patients with known esophageal disorders as there is evidence that it prevents and improves esophageal disorders [9][10]. Etiologies include scleroderma, achalasia, and malignancy. Symptoms suggestive of esophageal dysmotility are variable and range from regurgitation, epigastric pain, and sensation of food being retained in the esophagus. The dysmotility our patient experienced was likely related to mass effect from her abscess on the gastroesophageal junction and gastric pouch. Management involved treatment of the underlying disease process, as evidenced by the resolution of the patient’s symptoms after surgical drainage of her abscess. Although not supported radiographically, the patient’s clinical course and microbiologic evidence was highly suspicious for occult anastomotic leak. Anastomotic leak is a feared complication of gastric bypass, carrying a mortality rate of nearly 10%, but occurs in less than 0.8% of cases within the first 30 postoperative days [11]. Leakage of gastric contents from an anastomotic site can lead to contained infection/abscess or even lead to diffuse peritonitis. Signs of leak traditionally manifest within the first postoperative week, and vary in presentation from tachycardia, tachypnea, and abdominal pain. Our patient’s leak demonstrated an uncommon presentation, given that occurred 12 days post Roux-en-Y and was accompanied with evidence of esophageal dysmotility. Instead of overt signs of peritoneal irritation or infection, the patient had a normal white count and was afebrile without tachycardia or tachypnea. Given the timing of these events and absence of radiographic and intraoperative evidence of anastomotic leak throughout the case, it is reasonable to believe that an increase in intraluminal pressure during complete obstruction caused tension on the anastomosis leading to transient failure of the junction and subsequent seeding of gastric contents into the lesser sac [12]. Due to the limited length of her obstruction and surgical intervention, the path of least resistance again became the Roux limb and the leak sealed spontaneously. Management of leak includes aggressive evaluation for signs and symptoms of early sepsis, imaging with CT or upper gastrointestinal study, and diagnostic laparoscopy if tests are inconclusive [2]. In our patient, operative abscess evacuation with drain placement, anaerobic antibiotic coverage, and G-tube placement for early feeding were important components of care. Ironically, treatment of this patient’s leak began with her adhesiolysis performed seven days prior to her current presentation. | ||||||
| ||||||
|
CONCLUSION
| ||||||
|
In conclusion, this patient initially presented with a transient small-bowel obstruction resulting from an adhesion, leading to a transient increase in intraluminal pressure. We theorize that this resulted in a brief disruption of the anastomotic site, which then progressed to an abscess. Interestingly, instead of appearing with an acute infectious process, the patient presented with esophageal dysmotility. In discussing this case, we illustrate a unique constellation of events following Roux-en-Y gastric bypass surgery. | ||||||
|
REFERENCES
| ||||||
| ||||||
| [HTML Abstract] [PDF Full Text] |
|
Acknowledgements
We thank Northwest Hospital and LifeBridge Health for their support in this paper. |
|
Author Contributions
Ryan D. Nicklas – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Daniel S. Swink – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Alyssa E. Heinrich – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Christopher Boyd – Substantial contributions to conception and design, Acquisition of data, Revising it critically for important intellectual content, Final approval of the version to be published Christina Li – Substantial contributions to conception and design, Acquisition of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2017 Ryan D. Nicklas et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|