|
 |
|
Case Report
| ||||||
| Aneurysmal changes in silent patent ductus arteriosus in a child (year time follow-up) | ||||||
| Aso F Salih1, Rozhin O. Qadir2 | ||||||
|
1Department of Pediatrics, School of Medicine, Faculty of Medical Sciences, University of Sulaimani, al-sulaymanyah, Iraq
2M.B.Ch.B, Pediatric teaching hospital, al-sulaymanyah, Iraq | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article] [Similar article in Pumed] [Similar article in Google Scholar] 
|
| How to cite this article |
| Salih AF, Qadir RO. Aneurysmal changes in silent patent ductus arteriosus in a child (year time follow-up). Int J Case Rep Images 2017;8(11):711–716. |
|
ABSTRACT
| ||||||
|
Ductus arteriosus aneurysm is a saccular or fusiform dilation and elongation of the patent ductus arteriosus (PDA). It can be either congenital or acquired. Their time of presentation can be before or after treatment. In our case, aneurysmal found during her follow-up with cardiologist, it shows itself after a cardiac catheterization for trail of closure after 15 months from first catheterization. So decision was made for surgical closure rather than trial of occlusion with occluder devices. Surgery was performed after nearly one month from catheterization, under general anesthesia and median sternotomy, cardiopulmonary by-pass without complications. In conclude, duct aneurysm may happen in any type of PDA even very restrictive one and should be treated affectively. We conclude that although PDA relatively is common and safe heart defect but they may complicate further with age, even restrictive PDA may complicate by aneurysmal PDA if left untreated and it should be consciously treated. Keywords: Aneurysm, Catheterization, Patent ductus artriosus | ||||||
|
INTRODUCTION
| ||||||
|
Patent ductus arteriosus (PDA) is a vascular communication between descending thoracic aorta distal to the origin of the left subclavian artery and main pulmonary artery near the origin of the left pulmonary artery, it is an abnormal persistency of the fetal ductus arteriosus, which is an essential fetal structure, that should close spontaneously in about 90% of full-term infants during the first 48 hours after birth. It is persistency beyond a few weeks is considered abnormal [1]. It may be an isolated or occur with other congenital cardiac anomalies. Ductus arteriosus aneurysm (DAA) is a saccular or fusiform dilation and elongation of the PDA [2]. It can be either congenital or acquired as a complication of surgical closure of PDA or after a ductal infection [3] [4]. Their time of presentation can be divided in two three groups according to the age at diagnosis: infantile < 2 months, childhood (2 months to 15 years) and adult [5]. DAA has been considered as a rare congenital anomaly with potential fatal complications [5][6][7][8], although the exact incidence of DAA is unclear, but it can be ranged between 0.8–8% among fetuses and neonates [7][9], and approximately 5.2% of all adult thoracic aneurysm [10], with some reported childhood cases [3][11][12]. Some authors believe that neonatal DAA is resulted from transient widening of the ductus arteriosus prior to spontaneous obliteration and are not true aneurysm [5][13][14]. So the incidence of true DAA may be lower than previously believed [15]. Here we report a five-year-old child with PDA that was diagnosed incidentally at age four years, during second angiographic trial of closure found to have a DAA (pulmonary end aneurysmal), successful closure done with surgery. | ||||||
|
CASE REPORT
| ||||||
|
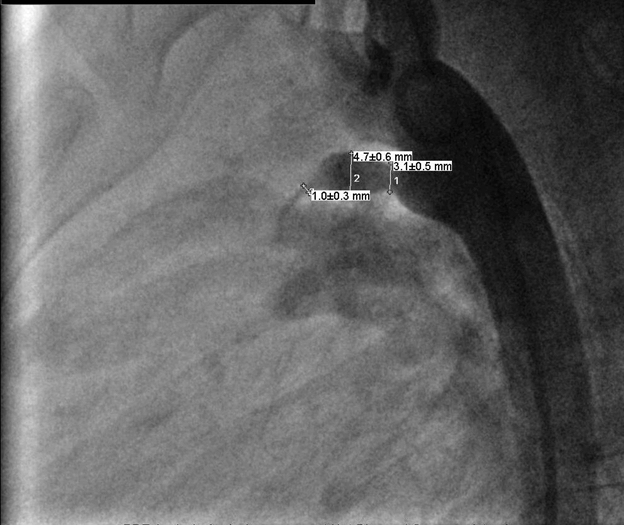
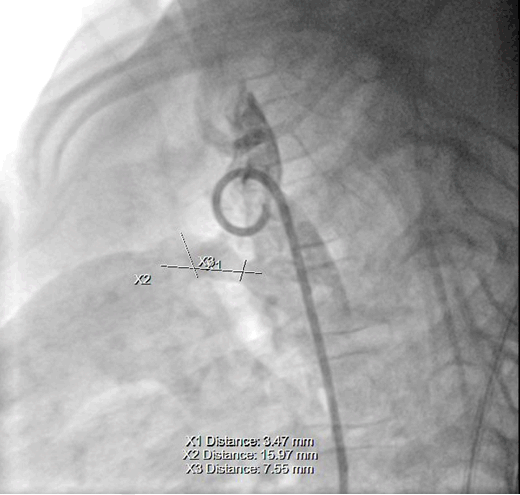
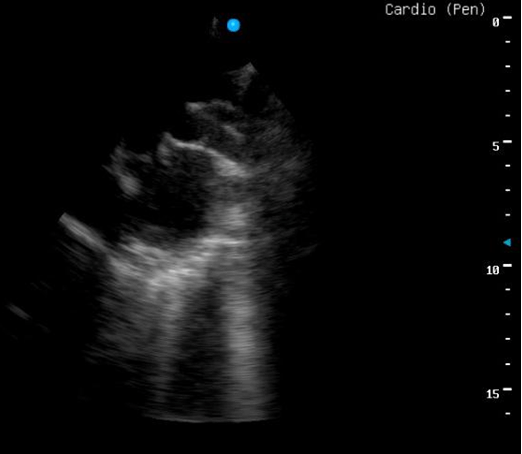
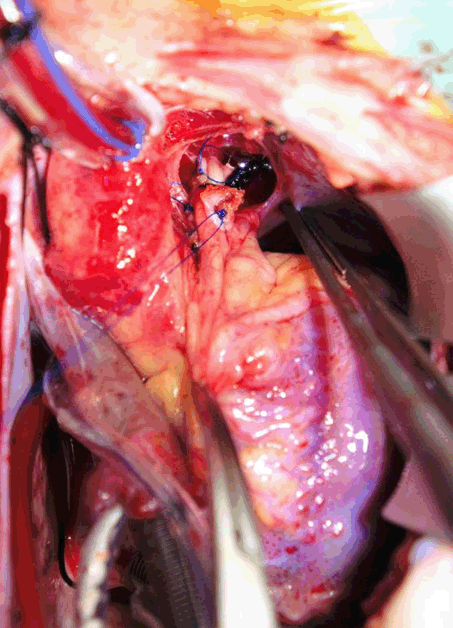
A five-year-old girl with history of premature birth and normal vaginal delivery, since then she was completely normal and had regular follow-up with her pediatrician. During routine checkup at age four year, found to have abnormal heart sound on physical examination of the chest by her pediatrician without aving any signs and symptoms. Then she was referred to pediatric cardiologist, found to have a non-restrictive medium sized patent ductus arteriosus (3.3 mm) with mild left ventricular enlargement, trivial MR and mildly increased pulmonary pressure by transthoracic echocardiography without evidence of any other congenital heart anomaly. On physical examination, she was normal for her height and weight, no any signs of respiratory distress, no cyanosis and no clubbing. Vital signs were normal. On chest examination no any deformity found, no thrill, with grade three continuous machinery murmurs in the left upper sternal boarder. Both electrocardiography and chest radiography were normal. The patient was arranged to have a cardiac catheterization with trial of closure of the PDA if possible, her PDA was type D subtype (complex duct with multiple constrictions) with aortic side is 3.1 mm *midpoint 4.7 *pulmonary end 1.0 mm (Figure 1) but because of unavailability of occluder devices, closure was postponed. During her follow-up with cardiologist, no any additional echocardiographic change was noticed. Only during physical examination found to have grade 4 thrill with radiation to the neck. She was arranged for another cardiac catheterization for trail of closure once occluder devices became available in our center after 15 months from first catheterization. During the second catheterization aneurysmal change seen at the pulmonary end rather than aortic end of the PDA with aortic end 3.4 mm* midpoint 7.5 mm* pulmonary end 15.9 mm (Figure 2), so decision was made for surgical closure rather than trial of occlusion with a occluder devices (Figure 3). Surgery was performed after nearly one month from catheterization, under general anesthesia and median sternotomy, cardiopulmonary by-pass without cardioplegia or circulatory arrest. Aneurysm found at the pulmonary end, simple ligation with non-absorbable suture material then aneurysmorrhaphy done at both pulmonary and aortic end with a pericardial pledged, smooth postoperative period without any complication. Immediate postoperative echocardiography showed trivial shunt across the PDA but with a week later no shunt was seen across the PDA anymore (Figure 4). | ||||||
| ||||||
| ||||||
| ||||||
| ||||||
|
DISCUSSION
| ||||||
|
The incidence of isolated PDA accounts for 9–12% of all CHD with higher proportion for female, with a female to male ratio 2:1 [16][17]. Postnatal closure of PDA occurs in two stages. First stage with the contraction of the medial smooth muscle in the wall of the ductus that leads to shortening and increased wall thickness. There is also protrusion of intimal cushions into the lumen [18][19]. These lead to functional closure of the ductus that take place within 12–15 hours after birth in full term infant. Second stage within 2–3 weeks after birth there is infolding of endothelium and proliferation of sub intimal layers which leads to fibrosis and permanent sealing of the lumen to produce ligamentum arteriosum [20]. The size of the PDA (length and the size of the ampulla) can vary from few millimeters to several centimeters, it may enlarge with age [21]. The PDA can be classified according to their size of the internal ductal diameter and shapes on lateral angiographic views. PDA is silent if it is less than 1 mm, very small if less than or equal to 1.5 mm, small if it is 1.5 to 3 mm, moderate if it is 3–5 mm and large if it is more than 5 mm [22]. The angiographic appearance of isolated PDA can be classified into five types that has been described by Kirchinko in 1989 [23]: Type A or conical type of PDA has a narrow end at the pulmonary insertion with a well-defined ampulla at the aortic end. Type B or window type of PDA is short and wide it may be narrow at aortic insertion. Type C or tubular type of PDA is tubular without any constriction. Type D or complex type of PDA has multiple constrictions (like our patient). Type E elongated type of PDA has a bizarre shape with an elongated conical shape and the constriction been remote from anterior border of trachea. Type A and B are further classified into three subgroups according to their site of insertion at the pulmonary end to the tracheal shadow [23]. Usually, closure of ductus begins at pulmonary end [5][24], and may remain incomplete at aortic end leaving small diverticulum adjacent to aorta, which is regarded as a transient and physiological part of normal spontaneous closure of ductus arteriosus called ductus bump [25][26]. Delayed closure of aortic orifice of the ductus exposing ductal tissue to systemic pressure is the most likely pathogenesis of aneurysm formation [5][24]. In our patient aneurysmal change was noticed at the pulmonary end than aortic end that represent about 30% of neonatal cases of DAA [5], mostly in neonates with pulmonary hypertension [6], but in our patient (childhood type), because she had a mild pulmonary hypertension, we think the same pathophysiology can applied here too. However, this does not explain the fetal or early neonatal development of DDA, when pressure in pulmonary artery is equal or higher to that in the aorta. In fact, there are several other theories about pathogenesis of DDA. First, congenital wall weakness may result from necrosis and mucoid degeneration of the media in the ductal wall [5]. Second increased flow throws the ductus arteriosus in the uterus a concomitant exposure to an increased arterial pressure which occurs in the third trimester lead to aneurysmal formation [7][27]. Third although there is no evidence of ductal stenosis on the prenatal echocardiogram [6][7][15][28][29], but intrauterine ductal constriction may cause post-stenotic dilatation of ductus arteriosus [30]. Finally, abnormal elastic fiber and extracellular matrix deposition within the wall of ductus arteriosus may occur in the setting of connective tissue disorder like Marfan, Ehlars–Danlos and Larsen syndromes has been observed in some cases of DAA which may result in weakening of the wall of the ductus arteriosus [2][6]. None of these theories can explain the pathogenesis of DAA in all age groups. Even some papers consider DAA as normal variant of elongated ductal bump [9]. Previously, DAA was considered as rare cardiovascular lesions, mostly was described in isolated case report [2]. The most common age at diagnoses is within the first two months after birth [6], but the pathology can also be diagnosed prenatally [6] [7][15][28]. Sign and symptom of ductus arteriosus aneurysm are usually scarce and discrete. Majority of affected neonate with DAA usually clinically asymptomatic [6][9], and complication may occur but very rare in newborn with DAA, with more symptomatic patient having larger DAA diameter and more commonly associated with complications [9]. However even smaller DAA might be associated with fatal complications in patient with connective tissue disease [5]. There are report suggesting that congenital DAA that developed postnatally may be associated with a higher rate of complications especially those who are symptomatic [5][8][14][31][32]. They may present with wide spectrum of symptoms with or without complications like spontaneous rupture, thromboembolism, erosions and compression of the adjacent structures including airway and the recurrent laryngeal nerve and even death[5][6] [7][8][33][34][35]. Our patient was asymptomatic until age fourth year when she was examined by her pediatrician as a part of regular neonatal follow-up because of her prematurity which we think that the PDA was present initially and of silent type and have gone undetected, even though she was clinically asymptomatic but once the diagnosis was made the parents start to notice that their daughter having symptoms like easy fatigability and shortness of breath. The diagnostic modality for DAA after initial physical examination and chest X-ray is echocardiography which can provide a reliable and valuable method for both diagnosis and demonstration of DAA anatomy [9][12], there are three important signs on echocardiography including unusual ductal shunt jet, triple star sign and rabbit ear sign [9], and other imaging modality like CT angiography and MRI scan have also been used for diagnosis and the nature of DAA but they are rarely necessary unless there are clinical evidence of compression of extravascular structure [9]. But in this patient, although there were some echocardiographic changes in the nature of the PDA unfortunately was not diagnosed through serial echocardiographic follow-up until after the second trail for PDA closure through catheterization then we found that the PDA have gone through aneurysmal dilatation at the pulmonary end, probably such aneurysmal change was unexpected over this 14 months, rapidly checked and missed. In adult patient, a limited acoustic window may impair detail evaluation of this region by echocardiography and color Doppler may be unable to detect a DAA when there is no left-to-right shunting [36]. When we found that PDA was aneurysmal during the second catheterization decision was made to close the PDA surgically rather than trans-catheter closure because our patient had no high risk for surgery, apparently the application of these devices may be associated with later complications of the aneurysm that mandate re-operative surgery, which carries a highest mortality rate 26%, and without intervention mortality reaches 91% due to rupture and infection [4]. Our patient had a clear indication for operation rather than watchful waiting despite that she was completely asymptomatic. From our review of literature we found that surgical resection for DAA should be proposed if any of the following condition existed: first, patent ductus arteriosus with DAA persist beyond the neonatal period (our patient); second, DAA associated with connective tissue diseases; third: there is evidence of thrombus extension into adjacent vessels or thromboembolism and finally, significant compression of adjacent structures [6][9]. From our review of literature, we found that most of surgical resection of DAA with or without complications was carried out through median sternotomy with cardiopulmonary bypass machine[7], separate perfusion of the arch and the lower extremities [3] , and circulatory arrest [33]. and method of closure were hand sewing aneurysmorrhaphy [37] that has been performed in our patient or stapling in case of uncomplicated ductus aneurysm having a narrow neck [37] or with the help of a Gott shunt bypassing the aneurysm and patching the neck of aneurysm from inside the aorta [8]. In our case, both aortic and pulmonary end aneurysmorrhaphy with pledged from pericardium done without resection of the PDA after putting the patient on cardiopulmonary bypass as a stand by for any undesired complication intraoperatively. Simple ligation of PDA in case of DAA is contraindicated because of the likelihood of incomplete discontinuation of the flow and risk of rupture both intraoperatively and long-term [6]. Giving the likelihood of long-term complication that might happen with DAA even after complete surgical resection even without association of connective tissue diseases, these children need continued follow-up. | ||||||
|
CONCLUSION
| ||||||
|
We conclude that although PDA relatively is common and safe heart defect but they may complicate further with age, even restrictive PDA may complicate by aneurysmal PDA if left untreated and it should be cousiously treated. | ||||||
|
REFERENCES
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Aso F. Salih – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Rozhin O. Qadir – Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2017 Aso F. Salih et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|