|

|
 |
|
Case Report
| ||||||
| Late infantile metachromatic leukodystrophy in a two-year-old boy: A case report | ||||||
| Jillalla Narsing Rao1, Swathi Chacham2, Uppin Narayan Reddy3, Janampally Ravikiran4, S. Pratap Rao1, Battula Sugunakar Reddy5 | ||||||
|
1MD, Pediatrics, Professor, Department of Pediatrics, Princess Esra Hospital, Deccan College of Medical Sciences, Hyderabad, Telangana, India.
2MD, Pediatrics, DM, Neonatology (from PGIMER), Assistant Professor, Department of Pediatrics, Princess Esra Hospital, Deccan College of Medical Sciences, Hyderabad, Telangana, India. 3MD, Pediatrics, Professor and Head, Department of Pediatrics Princess Esra Hospital, Deccan College of Medical Sciences, Hyderabad, Telangana, India. 4MBBS, (MD, Pediatrics), Junior Resident, Department of Pediatrics, Princess Esra Hospital, Deccan College of Medical Sciences, Hyderabad, Telangana, India. 5MD, Pediatrics, Assistant Professor, Department of Pediatrics Princess Esra Hospital, Deccan College of Medical Sciences, Hyderabad, Telangana, India. | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] 
|
| How to cite this article |
| Rao JN, Chacham S, Reddy UN, Ravikiran J, Rao SP, Reddy BS. Late infantile metachromatic leukodystrophy in a two-year-old boy: A case report. Int J Case Rep Images 2015;6(4):228–232. |
|
Abstract
|
|
Introduction:
Metachromatic leukodystrophy (MLD) occurs due to cerebroside sulfatide accumulation in the extraneural and neural tissues. Arylsulfatase A (ARSA) enzyme deficiency leads to progressive focal or generalized demyelination. This rare disorder involves both central and peripheral nervous system.
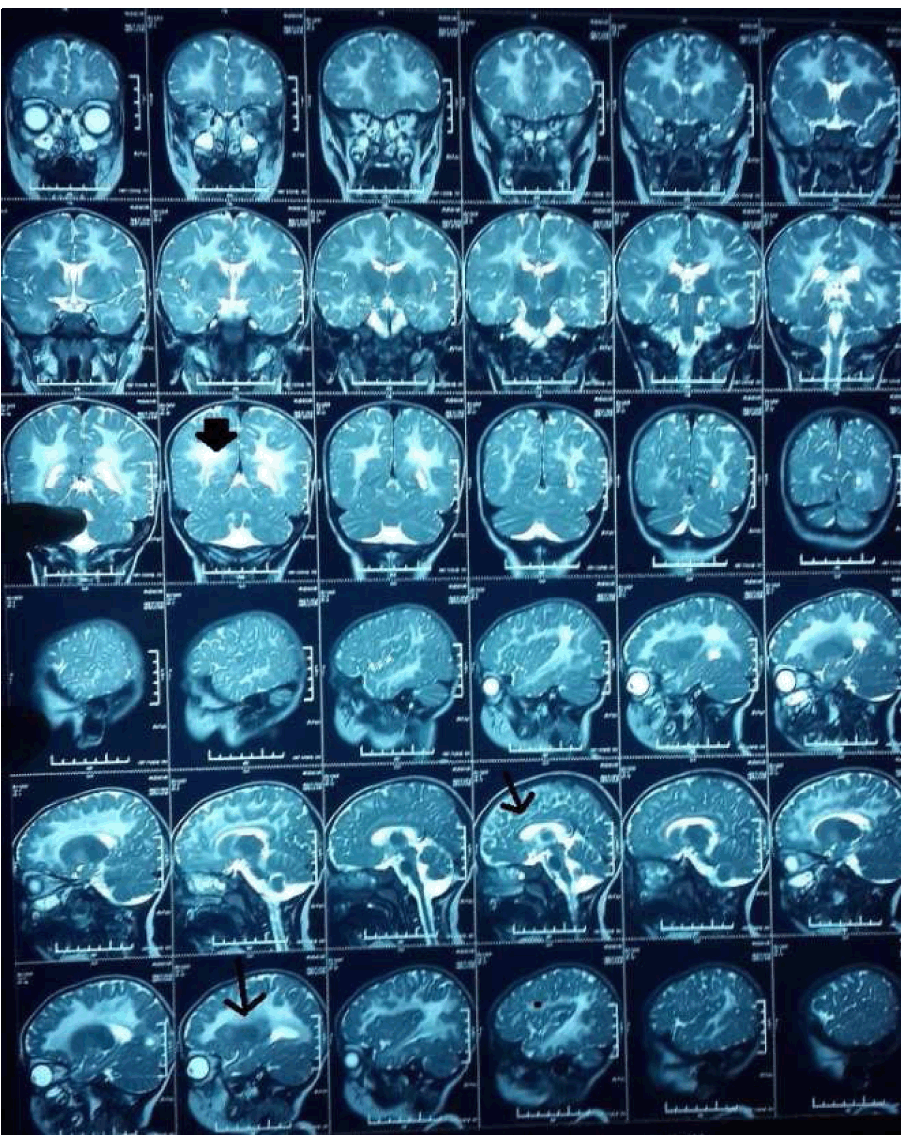
Case Report: We report a two-year-old boy, born of consanguineous marriage presenting with recurrent seizures from 13 months of age, followed by regression of milestones and spasticity from 15 months of age. Neurodegenerative disorder was considered initially and the child underwent neuroimaging followed by enzyme level assay. Magnetic resonance imaging scan (MRI) of brain revealed demyelination involving both cerebral cortex in the periventricular white matter with sparing of subcortical 'u' fibers and tigroid appearances /leopard skin sign, hallmark of metachromatic leukodystrophy. Normal β-galactosidase enzyme activity along with undetectable (ARSA) enzyme levels confirmed the diagnosis of late infantile variant of metachromatic leukodystrophy. Conclusion: A two-year-old boy presented with recurrent, generalized seizures, regression of milestones along with characteristic MRI findings and untraceable ARSA activity suggesting late infantile metachromatic leukodystrophy. | |
|
Keywords:
Arylsulfatase deficiency, Degenerative disorder, Infantile, Metachromatic leukodystrophy
| |
|
Introduction
| ||||||
|
Metachromatic leukodystrophy (MLD), a rare neurodegenerative metabolic disorder occurs with an incidence of 1 in 40, 000 to 1,60 000 individuals, worldwide [1]. Arylsulfatase A (ARSA) (a lysosomal enzyme) deficiency results in sulfatide accumulation within the myelin sheath of the central and peripheral nervous system, leading to progressive focal or generalized white matter degeneration [2] [3]. | ||||||
|
Case Report
| ||||||
|
A two-year-old male child born of third-degree consanguinity, presented with the history of generalized, recurrent seizures from 13 months of age. The developmental milestones were normal till one year of life. The developmental age in all quadrants (motor, language and social) was appropriate. The child had attained neck holding at third month and was able to sit with support by sixth month. Sitting without support was attained by eighth month and was walking with support by nineth months. At the age of one year, the infant was able to stand without support. The child was speaking mono-syllables by sixth month and bi-syllables by nine months. He showed stranger anxiety by 10th month. At the age of 13th month, the child developed generalized, tonic-clonic seizures which were associated with fever and minor respiratory illness. Seizures lasted for 20–25 minutes and required administration of two anti-epileptic agents. At the time of admission, there were no meningeal signs and CSF analysis (WBC 3 cells/mm3, neutrophils nil, glucose 56 mg/dL, protein 21 mg/dL, chloride 121 mmol/l, LDH 158U/l, ADA 4U/L, negative for acid-fast bacilli, culture and sensitivity- no bacterial growth) was not suggestive of meningitis. Hence the child was discharged on two anti-epileptic agents. At the age of 15th months, he developed recurrent seizure episodes, which necessitated administration of 3rd anti-epileptic agent. This episode was succeeded by regression of milestones which was progressive in nature. He developed progressive inability to walk, sit and speak with in a time span of one to two months. The child required frequent hospitalization for recurrent seizures and required multiple anti-epileptic agents. The antenatal period was uneventful and the infant was delivered by spontaneous vaginal delivery with birth weight of 3 kg. No history of perinatal asphyxia, neonatal seizures and neonatal jaundice. No maternal history of fever, rash in the antenatal period, which could be suggestive of toxoplasmosis, rubella, cytomegalovirus, and herpes simplex (TORCH) infection. No family history of similar complaints. On examination, there were no neurocutaneous markers and no facial dysmorphism. Head circumference was 46 cm. Cardiovascular system and respiratory system were unremarkable. However, central nervous system revealed generalized spasticity, exaggerated deep tendon reflexes and extensor plantar response. Retinal examination was unremarkable. Initial differential diagnosis of infantile stroke, cerebral palsy, and neurodegenerative disorder was considered and the child was further investigated. MRI scan of brain was abnormal and the β-galactosidase levels were normal. However, undetectable arylsulfatase A activity in the child confirmed the diagnosis of MLD. MRI brain image showed Tigroid appearances /Leopard Skin sign, suggesting metachromatic leukodystrophy(as shown with arrows in Figure 1). There was demyelination of periventricular white matter of cerebral cortex on both sides along with sparing of subcortical 'u' fibers(as shown in Figure 2). The patient was treated with supportive care together with physiotherapy. However, bone marrow transplantation, a newer modality of treatment was not feasible due to financial constrains. | ||||||
| ||||||
| ||||||
|
Discussion
| ||||||
|
Aryl sulfatase A, a lysosomal enzyme aids in degradation of sulfated glycolipids, especially galactosyl sulfatide. Classical type MLD results from this arylsulfatase A enzyme deficiency, which leads to accumulation of sulfatide in central and peripheral nervous system, progressive demyelination, motor and cognitive dysfunction. MLD has been categorized depending on the age of presentation as late-infantile type (onset before three years of age), juvenile type (onset before 16 years) and adult type [4]. Late infantile and the juvenile variants are characterized by rapid motor decline, while adult form presents with cognitive and behavioral problems. The index case is considered as late infantile variant of MLD, based on the age of onset and clinical presentation. The index child presented with typical regression of milestones following refractory seizures. Another characteristic feature of MLD is recurrent seizures and their incidence increases with duration of illness, as in the index child. Generalized seizures are common in infantile type (as in the present case), while partial seizures are more common in juvenile type. Older children present with gait abnormalities, seizures, behavioral changes and deterioration of scholastic performance. Along with regression of milestones, spasticity (a characteristic manifestation of MLD) was also present in the index child [5] [6]. The important diagnostic modalities used to confirm this degenerative disorder are arylsulfatase A enzyme activity, molecular genetic testing of arylsulfatase A, estimation of urinary sulfatide and detecting metachromatic lipid deposits in the nervous tissue [7]. Gene sequence analysis of arylsulfatase A is an important tool for prenatal diagnosis. As MLD progresses with age and the neurodegeneration worsens with time, there is no definitive treatment till date. Newer treatment modalities include stem cell transplantation, bone marrow transplantation along with genetic engineering and these might halt the progression of neurologic dysfunction [8] [9] [10]. Recombinant human ARSA administration, an experimental treatment can be a promising option in future, although it lacks universal recommendation and adaptation. | ||||||
|
Conclusion
| ||||||
|
A two-year-old boy born of consanguinity, manifesting with generalized refractory seizures followed by regression of milestones and spasticity. Characteristic MRI findings, tigroid appearance/leopard skin sign coupled with undetectable arylsulfatase A activity suggests late infantile variant of metachromatic leukodystrophy. | ||||||
|
Abbreviations
| ||||||
|
ARSA (Aryl sulfatase A), MLD (Metachromatic Leukodystrophy), MRI (Magnetic resonance imaging), TORCH (Toxoplasmosis, Rubella, Cytomegalovirus and Herpes simplex) | ||||||
|
Acknowledgements
| ||||||
|
We very much appreciate the assistance Dr madhavinoori and Dr Anumulasoumya for their help and support. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Jillalla Narsing Rao – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Swathi Chacham – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Uppin Narayan Reddy – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Janampally Ravikiran – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published S. Pratap Rao – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Battula Sugunakar Reddy – Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2015 Jillalla Narsing Rao et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|
|
About The Authors
| |||
| |||
| |||
| |||
| |||
| |||
| |||