|

|
 |
|
Case Report
| ||||||
| Stent thrombosis after switch from nongeneric to generic clopidogrel | ||||||
| George Stoupakis1, Louis Teichholz2 | ||||||
|
1MD, Associate Physician, Heart and Vascular Institute, Hackensack University Medical Center, Hackensack, New Jersey, USA.
2MD, co-Chairman, Heart and Vascular Institute, Hackensack University Medical Center, Hackensack, New Jersey, USA. | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] 
|
| How to cite this article: |
| Stoupakis G, Teichholz L. Stent thrombosis after switch from nongeneric to generic clopidogrel. International Journal of Case Reports and Images 2014;5(1):41–44. |
|
Abstract
|
|
Introduction:
The U.S. Food and Drug Administration recently approved generic versions of clopidogrel bisulfate which should lower the cost and improve compliance in patients requiring dual antiplatelet therapy post stent placement. However, there is marked inter-individual variability in the metabolism and intestinal absorption of clopidogrel. The different binders and fillers used in the generic versions can possibly affect absorption and reduce the bioavailability of the drug.
Case Report: We report a case of very late stent thrombosis in a patient eight days after the switch to generic clopidogrel bisulfate from branded clopidogrel (Plavix). The patient had received a sirolimus drug eluting stent almost three years prior to presentation and was on chronic dual antiplatelet therapy. Subsequent to the event, she was found to be a poor metabolizer of clopidogrel. Conclusion: Although in most cases the use of generic clopidogrel is efficacious and safe, certain individuals, such as poor metabolizers, might be susceptible to increased clinical events when switching. We suggest any suspected cases should be reported to Medwatch so the clinical relevance could be determined. | |
|
Keywords:
Stent thrombosis, Antiplatelet therapy, Clopidogrel
| |
|
Introduction
| ||||||
|
Clopidogrel is a thienopyridine which is orally administered in combination with aspirin to reduce the incidence of ischemic cardiovascular events in patients who undergo percutaneous coronary intervention (PCI). The U.S. Food and Drug Administration recently approved generic versions of clopidogrel bisulfate which should lower the cost and improve compliance in patients requiring dual antiplatelet therapy post-PCI. However, there is marked inter-individual variability in the metabolism and intestinal absorption of clopidogrel. The different binders and fillers used in the generic versions can possibly affect absorption and reduce the bioavailability of the drug. We report the first known case of very late stent thrombosis temporally related to the switch from branded clopidogrel (Plavix) to generic clopidogrel. | ||||||
|
Case Report
| ||||||
|
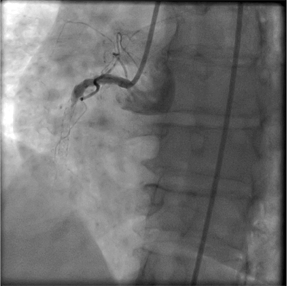
A 60-year-old female with a past medical history of myocardial infarction requiring stenting of the mid right coronary artery (RCA) with a Sirolimus drug eluting stent in 2009, presented to our emergency room in 2012, 1053 days after stent placement, with chest pain and diaphoresis and a new acute inferior wall ST-segment elevation myocardial infarction. Her electrocardiogram revealed a new 2- mm ST elevations in the inferior leads. (Figure 1) She was taken directly to the cardiac catheterization lab where she was found to have very late stent thrombosis of the mid RCA (Figure 2) and underwent successful thrombus aspiration and revascularization. (Figure 3) At the time of presentation, the patient was on chronic dual antiplatelet therapy with aspirin 325 mg daily and generic clopidogrel bisulfate 75 mg daily. It was confirmed, 8 days prior to this event, the patient was switched from original branded clopidogrel bisulfate (Plavix, Bristol Meyers Squibb/Sanofi) to generic clopidogrel bisulfate (Teva Pharmaceutical Industries Ltd.). The patient was a former smoker and was known to be compliant with her medications and office visits. She clearly stated that she did not miss any doses of her antiplatelet therapy. She was not taking proton pump inhibitors. Subsequent to the event, clopidogrel 2C19 genotyping found her to be a poor metabolizer. She was discharged from the hospital on aspirin 325 mg daily and prasugrel 10 mg daily. | ||||||
| ||||||
| ||||||
| ||||||
|
Discussion
| ||||||
|
On 17th May 2012, the U.S. Food and Drug Administration approved generic versions of clopidogrel. Generic medications reduce the overall cost of medical care and the introduction of lower-cost generic clopidogrel might increase patient compliance and reduce premature discontinuation of therapy. Although there are studies to show bioequivalence of formulations of clopidogrel in healthy adults, [1] [2] the ACCEL-GENERIC study is the only known prospective study which compared the degree of platelet inhibition between the original branded clopidogrel (Plavix) and a generic formulation of clopidogrel bisulfate in patients with coronary stents. [3] In this small study of 20 patients, all patients were treated with drug-eluting stents (DES) and received Plavix 75 mg daily for 6 months before being switched to generic clopidogrel. Platelet function measured at baseline and 30 days after generic clopidogrel replacement by light transmittance aggregometry (LTA) and the VerifyNow P2Y12 assay (Accumetrics, San Diego, CA, USA), demonstrated poor inter-therapy agreements between these two methods. Although there were no reported clinical events, because of this poor inter-therapy agreement, the authors concluded that there might be a risk of ischemic events in some patients when switching to generic clopidogrel. Furthermore, the authors stated that physicians should be cautious when routinely switching to generic clopidogrel due to cost, particularly during the early phase of coronary stenting or Acute coronary syndromes (ACS). [3] There is marked inter-individual variability in the metabolism of clopidogrel. Based on available data, the prevalence of clopidogrel resistance is estimated at 4–30% of patients treated with conventional doses of clopidogrel. [4] Mechanisms that affect the efficacy of clopidogrel include drug-drug interactions, polymorphisms of the P2Y12 receptor gene, polymorphisms of the CYP3As, and baseline platelet reactivity. Inter-individual variability of the intestinal absorption of clopidogrel is also considered to be an important determinant of response variability to clopidogrel. [5] [6] Physiologic factors can also affect the absorption of clopidogrel. For example, pharmacokinetic studies in patients presenting with ST-segment elevation myocardial infarction demonstrate impaired bioavailability of clopidogrel due to impaired intestinal absorption influenced by the physiologic state of STEMI. [7] In addition, there is also marked intra-individual variability in the antiplatelet effect of different clopidogrel salt formulations. In a cross-over study of healthy volunteers treated with clopidogrel hydrogen sulfate (bisulfate) or clopidogrel besylate, some subjects were observed to respond less to the besylate salt but better to the hydrogen sulfate salt, while others had the reverse pattern. [8] Therefore, some individuals may be sensitive to changes in formulations and/or bioavailability. The use of generic medications in cardiovascular disease has been reviewed. [9] Generic medications are equivalent chemically to their brand-name counterparts in terms of active ingredients but may differ in features, such as pill color and shape, inert binders and fillers. [9] It is possible that the binder and filler used in our patient’s generic formulation could have affected the intestinal absorption and reduced the bioavailability of clopidogrel. The FDA evaluated 2070 human studies comparing the absorption of brand name and generic drugs. The average difference in absorption between the generic and brand names was 3.5%. [10] In drugs with a narrow therapeutic index (NTI), this difference in absorption can have clinical relevance. In addition, our patient’s STEMI, occurring eight days after the switch to the generic clopidogrel, coincides with the timing of washout of the original branded clopidogrel (Plavix). The recovery trial demonstrated that after discontinuation of clopidogrel, >75% of patients returned to baseline platelet reactivity by washout day 5 and > 90% by day 6. [11] In our patient, the time frame between switching to generic clopidogrel and the onset of the clinical event, suggest that there could have been less antiplatelet effect due to the washout of the original branded clopidogrel (Plavix) and possible lower bioavailability of the generic clopidogrel. Several factors most likely contributed to this patient’s episode of stent thrombosis. Firstly, she was genetically proven to be a poor metabolizer of clopidogrel. However, it is most likely not due to clopidogrel resistance alone. Since this event occurred almost three years after DES stent placement and eight days after the switch to generic clopidogrel, it appears that the branded clopidogrel (Plavix) was effective in preventing stent thrombosis. Current bioequivalence criteria, as proposed by the Food and Drug Administration (FDA), require a 90% confidence interval for the ratios of the geometric means (generic : reference) of the AUC and Cmax to fall between 0.8 and 1.25 (80–125%). The use of the 80–125% reference range has been criticized for drugs with a narrow therapeutic index because a small change in systemic concentration of these drugs can lead to a markedly different therapeutic response or even toxicity. [12] The narrow therapeutic index of clopidogrel may be an additional factor, as a reduced bioavailability of generic clopidogrel and a concurrent change in concentration may have rendered the drug less effective. In our patient, because she was found to be a poor metabolizer, her platelet inhibition on the branded clopidogrel (Plavix) might have been on the lower level of effectiveness, and the change to generic clopidogrel, with the possibility of less bioavailability, tipped the balance towards thrombosis. However, since platelet function studies were not performed on our patient before or after the switch to generic clopidogrel, we do not have objective measurements to support our hypothesis. It’s a speculation that the late thrombosis is most likely secondary to changing to generic clopidogrel, after three years of using Plavix, and not an incident event. | ||||||
|
Conclusion
| ||||||
|
We believe that, in general, the use of generic clopidogrel in most cases will be efficacious and safe. However, in some individuals there may be a variable response in bioavailability which could alter the efficacy, especially in those who are poor metabolizers. This is the first known case of very late stent thrombosis temporally related to the switch from branded clopidogrel (Plavix) to generic clopidogrel. Therefore, physicians should have an awareness of a potential for clinical events when switching to generic clopidogrel. We suggest any suspected cases to be reported to Medwatch. If this is more than just an isolated case, it might be prudent to make an assessment of genotyping, platelet function, lesion complexity and clinical risk factors in patients when considering a switch to generic clopidogrel. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
George Stoupakis – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Louis Teichholz – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© George Stoupakis et al. 2014; This article is distributed the terms of Creative Commons attribution 3.0 License which permits unrestricted use, distribution and reproduction in any means provided the original authors and original publisher are properly credited. (Please see Copyright Policy for more information.) |
|
|