| Table of Contents |  |
|
Case Series
|
| Case series of visceral larva migrans in the liver: CT and MRI findings |
| Shalini Thapar Laroia1, Archana Rastogi2, Shiv Sarin3 |
|
1Assistant Professor, Radiology department, Institute of Liver & Biliary sciences, New Delhi, India.
2Associate Professor, Department of Hepato Pathology, Institute of Liver & Biliary sciences, New Delhi, India. 3Director ILBS and Professor of Hepatology and Transplant Hepatology, Institute of Liver & Biliary sciences, New Delhi, India. |
|
doi:10.5348/ijcri-2012-06-129-CS-2
|
|
Address correspondence to: Dr. Shalini Thapar Laroia L-1 Krishan Vatika DLF Westend Farms, Govind Sadan Marg Chattarpur, New Delhi-110030 India Phone: 091-9810757973, 091-9811015080 Fax: 091-011-26806356 Email: thaparshalini@gmail.com |
|
[HTML Abstract]
[PDF Full Text]
|
| How to cite this article: |
| Laroia ST, Rastogi A, Sarin S. Case series of visceral larva migrans in the liver: CT and MRI findings. International Journal of Case Reports and Images 2012;3(6):7–12. |
|
Abstract
|
|
Introduction:
Visceral larva migrans (VLM) is an uncommon parasitic infection most commonly caused by Toxocara canis and Toxocara cati. Imaging features of hepatic VLM on contrast enhanced computed tomography (CECT) include presence of small, single or multiple (usually less than two cms in diameter) hypovascular eosinophilic abscesses seen enhancing only on portal venous phase (PVP) on triple phase liver study. Review of literature revealed that CT and peripheral eosinophilia in the blood has been the mainstay of clinical diagnosis in majority of case reports and larger studies.
Case Series: We have used 3 Tesla (3T) magnetic resonance imaging (MRI) in addition to CT for diagnosis of hepatic visceral larva migrans in three patients with special emphasis on T1 weighted and diffusion weighted sequences. MRI and CT images showed interesting findings such as hyperintense rim of the lesions on T1 weighted sequence and hyperintensity on diffusion weighted images (DWI) at b values of 1000 with associated restriction on the corresponding ADC maps. Conclusion: We would like to emphasize the importance of echo planar imaging and T1 weighted sequences of the liver in addition to routine dynamic CT and conventional MRI sequences for assessment of suspected eosinophilic abscesses in visceral larva migrans presenting as focal liver lesions. | |
|
Key Words:
Visceral Larva migrans, MRI, DWI, ADC, T1 weighted sequence, Eosinophilic abscesses
| |
|
Introduction
|
|
The second-stage larvae of nematodes (Toxocara is the commonest) are sometimes accidentally ingested by human beings and are released in the small bowel, from where they enter the portal venous system through the intestinal walls and infest the liver, lung, eyes etc. [1] [2] [3] imaging features of hepatic lesions in visceral lerva migrans (VLM) are indicative but not characteristic of the disease and need correlation with laboratory parameters like antigen serology, cytology and eosinophilic counts. However, with the help of triple phase CT, advanced MRI and newer sequences like DWI, imaging diagnosis can be made more specific. We present three cases with different clinical presentations who underwent triple phase CT in one case and MRI with diffusion weighted imaging (DWI) in two cases. They were diagnosed as eosinophilic (VLM) abscesses on imaging and proven with cytology and lab criteria. [12] The informed consent for the report was received from the patients and the ethics committee of our hospital. |
|
Case Series
|
|
Case 1: A 35-year-old female patient presented with intermittent high grade fever with chills for a duration of 1.5 years. She had been partially treated during past episodes of fever with antibiotics tazabactum and metronidazole. No definite history of contact with animals or pets was obtained. At the time of presentation, her total leukocyte counts were 7,400/mm3, eosinophil count was 4% and ESR was raised to 75 mm/1st hr. Hydatid serology was negative. Triple phase CT of the liver showed few coalescing, hypo-attenuating, solid, rounded lesions with minimal vascularity (Figure 1A-C) seen only on portal venous phase. Ultrasound guided FNAC of the liver lesion (Figure 1D) showed charcot leyden crystals with mixed inflammatory and proteinaceous material, predominant eosinophils and poorly formed epitheloid granulomas characteristic of eosinophilic abscesses of parasitic origin. Treatment with albendazole led to remarkable regression in size of these lesions at follow up. |
|
|
|
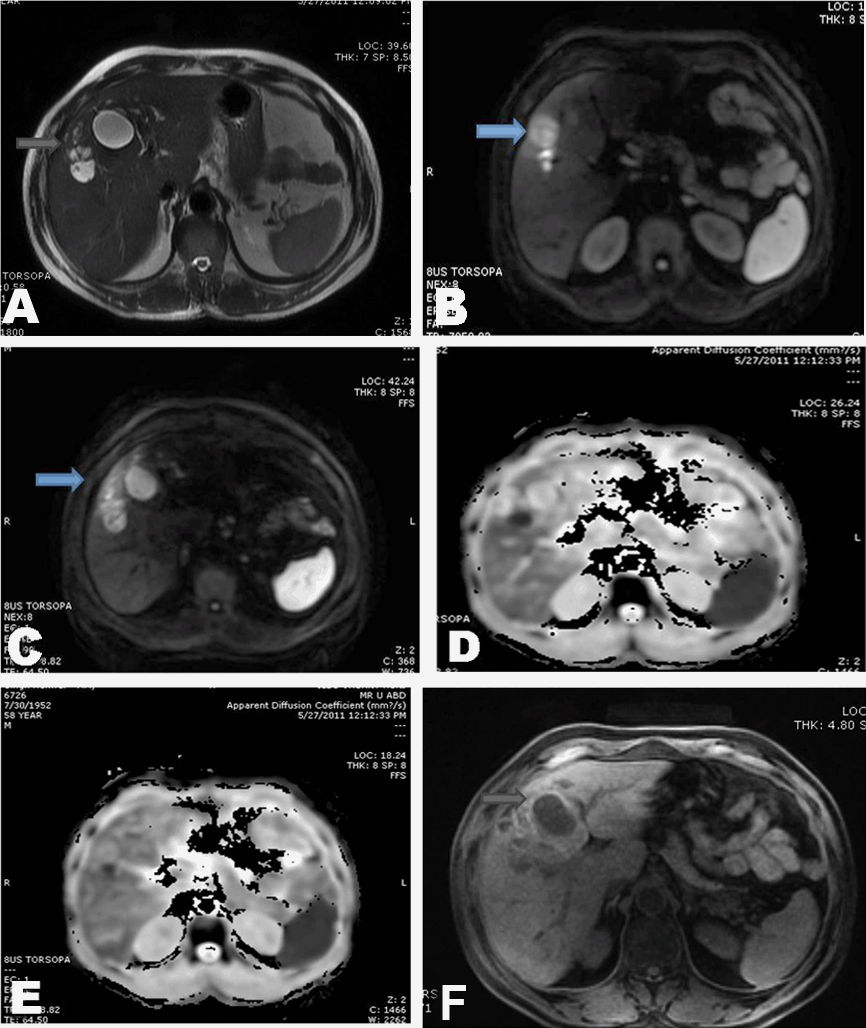
Case 2: Fifty-eight years male with low grade fever off and on for past three months was investigated and found to have raised total leukocyte count of 11,500/mm3, high ESR - 70 mm/1st hr, increased eosinophil count (38%) with altered liver enzymes and mildly raised total bilirubin of 2.5 mg/dl. His hydatid serology was negative. MRI was done for evaluation and multiple well defined thick walled T2 hyperintense lesions (Figure 2A) with brightness on diffusion weighted images (b value 1000) (Figure 2B, C) and corresponding restriction on ADC was seen (Figure 2D, E). In addition these lesions had T1 hyperintensity along their peripheries Figure 2F), possibly due to high cellularity. Parasitic eosinophilic abscesses due to VLM were diagnosed in correlation with peripheral eosinophilia. Anti-helminthic treatment showed significant decrease in lesion size and clinical symptoms. |
|
|
|
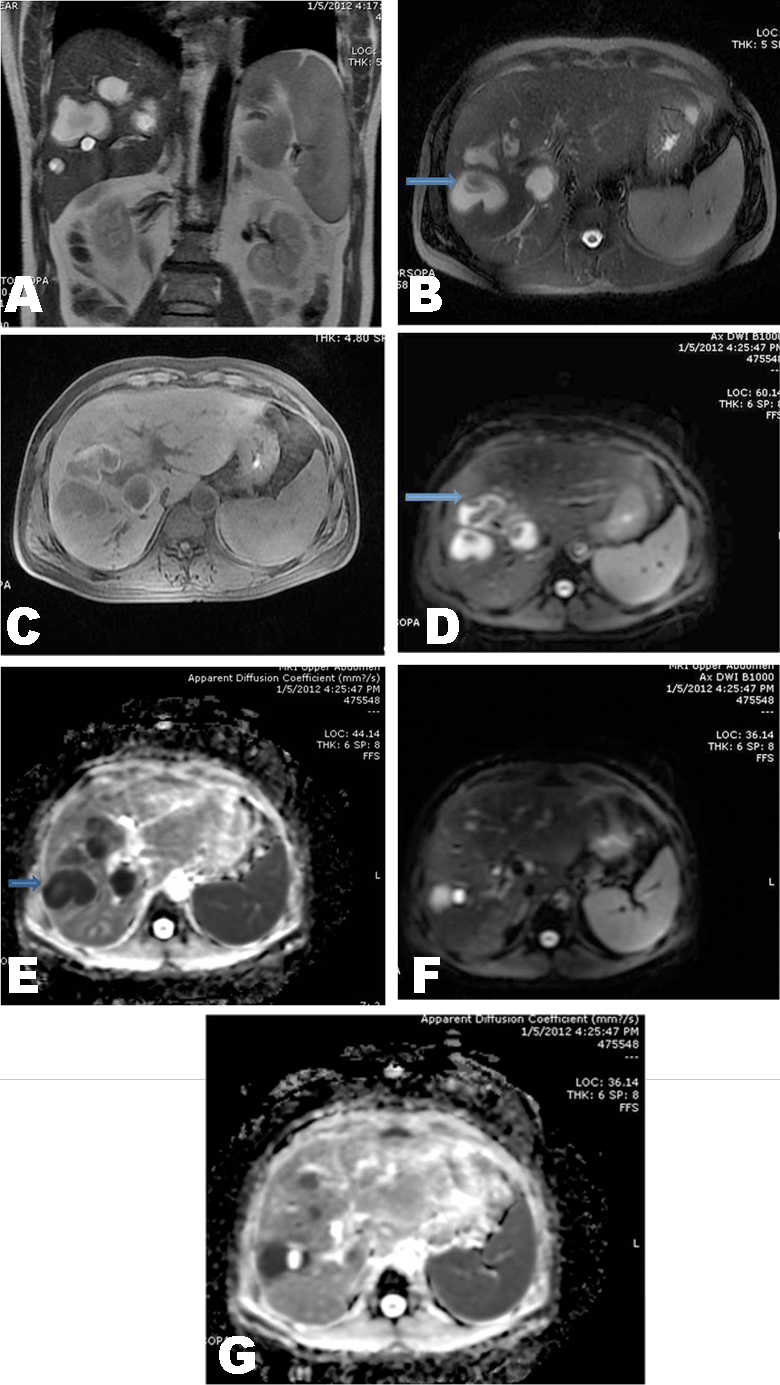
Case 3: Fifty one years male patient who was previously treated for non resolving atypical liver abscess was referred for an MRCP and upper abdominal scan to rule out biliary cystadenomas. His amebic and hydatid serologies were negative with marginally altered liver enzymes and raised bilirubin of 3.4 mg/dl. The total leukocyte counts were normal with mild eosinophilia of 8%. MRI scan showed conglomerating T2 hyperintense lesions with thick nodular regions along the walls (Figure 3A, B). T1 hyperintense rim of few of these lesions was noted (Figure 3C). On diffusion weighted sequence the abscesses showed hyperintensity at b value 1000 with corresponding restriction on ADC maps (Figure 3D-G). A diagnosis of parasitic eosinophilic abscesses was suggested and proven on ultrasound guided biopsy, which showed eosinophils and charcot leyden crystals with necrosis. |
|
|
|
Discussion
|
|
Visceral larva migrans is a systemic manifestation of toxocariasis. It is usually an asymptomatic or vaguely symptomatic condition which is caused by migrating larvae of Toxocara in the liver, lungs eyes etc. VLM has a worldwide prevalence, although there is a strong predilection for the tropical countries and southeast Asia. [1] The wide range of symptoms which are dependent on the organ involved, the intensity of larva load, infection and host immune response determine the course of the disease. Young adults and children who are in close contact with pets and animals or have increased exposure to public places and parks with contaminated soil are at a higher risk of contracting the disease. [4] [5] An unusual method of contracting this disease is through ingestion of raw infected meat containing the infective encapsulated larvae form of T. canis, reported by Ishibashi et al. [4]. None of the patients in the series presented above had history of animal contact or eating raw meat and hence clinical suspicion of the disease entity of parasitic origin could not be made. As the name VLM suggests, larval migration in organs such as liver and lung occurs, which causes reactive eosinophilic infiltration, parenchymal destruction, granuloma or eosinophilic abscess formation. [5] Eosinophilic abscess on histopathology shows enlarged eosinophils, necrosis, debris and destroyed liver architecture with infiltrates and inflammatory cells such as neutrophils and lymphocytes. [5] The larva may also be visualized rarely within tissue specimens. Imaging by ultrasound, CT and MRI is an attribute to these pathological findings. There have been series and case reports of VLM [2] [3] [4] [5] [6] [7] [8], and the radiologic findings have been described predominantly on ultrasound, CT and conventional sequences on MRI. Chang et al. described the CT and ultrasound findings of a retrospective analysis of 70 serologically proven hepatic VLM. [1] On contrast CT of the liver, lesions have been described as hypodense, non-subcapsular in location, usually multiple in number, ill-defined, oval angular or trapezoid lesions measuring 1.0–1.5 cm in diameter. [4] [6] [10] Different shapes and margins of these lesions depend on the type of destruction of the liver parenchyma by the abscesses. [10] They have well defined margins and walls with nodularity which can sometimes be seen on non contrast and dynamic triple phase MRI, studies. The portal venous phase best depicts this lesion nodularity and margin. Faint vascularity seen on arterial and equilibrium phases have been described by Azuma et al. and Kim et al. [9] [10] Characteristic T1 weighted hypointensity-isointensity and T2 weighted hyperintensity is documented by Kim et al. [9]. The differentiation from metastatic lesions is done by looking for fuzzy margins, elliptical shape and uniform sized lesions of 1–2 cm diameter. The conspicuity of the eosinophilic abscesses is most on the portal venous phase, which may later change size and positions, characteristic of migration of larva in the liver as opposed to metastases, supporting the diagnosis of VLM. [1] [10] [11] Some lesions have been seen to enhance on hepatic arterial phase but do not show any washout on equilibrium and delayed phases and hence can be differentiated from the nodules of hepato cellular carcinoma (HCC). [10] Differentiation from focal eosinophilic infiltrates is with the help of MRI and recently with the help of diffusion weighted imaging, which does not show any restriction of these benign lesions in the liver. [11] In the case studies presented above we analyzed the CT and MRI findings of eosinophilic abscesses of visceral larva migrans in patients with incidentally detected liver lesions, with hypereosinophilia, raised ESR and leucocyte count in two of them. We demonstrate the characteristic CT findings of VLM with a few additional interesting features on MRI that are helpful in differentiating them from other benign lesions, tumours and metastases in the liver. On contrast CT the lesions appeared coalescing, predominantly in the periportal and non-subcapsular region with well defined margins, hypoattenuation (approximately 35 HU) and well visualized on the portal venous phase. On non-contrast MR study, two kinds of imaging findings were shown: the first was the well defined hyperintense rim of the abscesses and the second was the restriction of these lesions on ADC maps corresponding to the diffusion images. Diffusion weighted sequences obtained at b values of 0 and 1000 on the 3Tesla MRI system demonstrated hyperintensity of the parasitic abscesses. Ahn et al. described the use of gadoxetic acid-enhanced MRI to diagnose focal eosinophilic infiltrates [11] in cases where lack of information from DWI sequencing of the focal infiltrates was shown. Their study group consisted of eight patients. 'Eosinophilic infiltrates' which they described has been proven to be a different entity from VML induced eosinophilic abscesses as described by Kim et al. [10] The presence of T1 hyperintense rim around the periphery of these lesions could possibly be due to increased cellularity in this region. To our knowledge there is no study in literature demonstrating the importance of these specific MR imaging characteristics in visceral larva of the liver. |
|
Conclusion
|
|
In conclusion, we would like to emphasize the importance of echo planar imaging of the liver in addition to routine dynamic CT and conventional MRI sequences for assessment of suspected eosinophilic abscesses in visceral larva migrans presenting as focal liver lesions. |
|
References
|
|
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions:
Shalini Thapar Laroia - Substantial contributions to conception and design, Analysis and interpretation of data, Drafting the article, revising it critically for important intellectual content, Final approval of the version to be published Archana Rastogi - Substantial contributions to conception and design, Drafting the article, Final approval of the version to be published Shiv Sarin - Substantial contributions to conception and design, Drafting the article, revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission:
The corresponding author is the guarantor of submission. |
|
Source of support:
None |
|
Conflict of interest:
Authors declare no conflict of interest. |
|
Copyright:
© Shalini Thapar Laroia et al. 2012; This article is distributed the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any means provided the original authors and original publisher are properly credited. (Please see Copyright Policy for more information.) |
|
|