| Table of Contents |  |
|
CLINICAL IMAGE
|
| Severe hepatic encephalopathy and decrease in enhancement of hepatocellular carcinoma treated with sorafenib |
| Takefumi Kimura1, 2, Takeji Umemura2, Tetsuya Ichijo2, Nobuyoshi Yamamura1, Eiji Tanaka2 |
|
1Department of Gastroenterology, Suwa Red Cross Hospital, Suwa, Japan; 2Department of Medicine, Shinshu University School of Medicine, Matsumoto, Japan.
|
|
doi:10.5348/ijcri-2010-12-12-CI-4
|
|
Address correspondence to: Takeji Umemura Department of Medicine Shinshu University School of Medicine 3-1-1 Asahi, Matsumoto Japan - 390-8621 Phone: +81-263-37-2634 Fax: +81-263-32-9412 Email: tumemura@shinshu-u.ac.jp |
|
[HTML Abstract]
[PDF Full Text]
|
| How to cite this article: |
| Kimura T, Umemura T, Ichijo T, Yamamura N, Tanaka E. Severe hepatic encephalopathy and decrease in enhancement of hepatocellular carcinoma treated with sorafenib. International Journal of Case Reports and Images 2010;1(4):16-18. |
|
Case Report
| ||||||
|
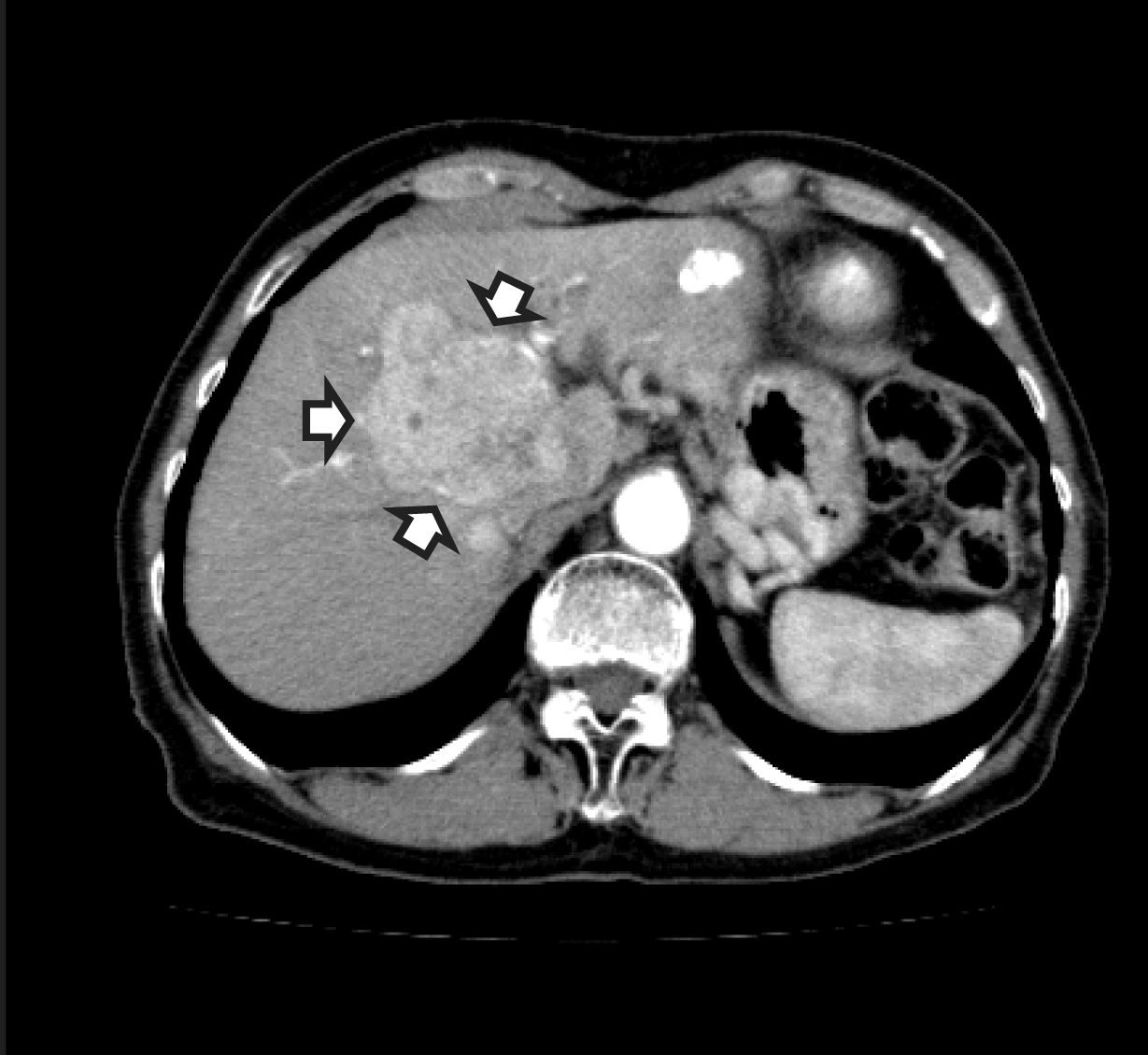
Sorafenib (Nexavar; Bayer Pharmaceuticals Group, Montville, NJ) is an oral multikinase inhibitor recently licensed in Japan for the treatment of advanced hepatocellular carcinoma (HCC) in May 2009. Although the most frequently reported adverse events of sorafenib have been fatigue, anorexia, diarrhea, rash, and hand-foot skin disease, [1] we here report a patient who developed severe hepatic encephalopathy that accompanied marked improvement of HCC after only three days of treatment with sorafenib. A 79-year-old woman with hepatitis C virus-related cirrhosis (Child-Pugh A; 5: prothrombin 78%; bilirubin 1.7 mg/dl; albumin 3.6 g/dl; no ascites or encephalopathy) had progressive multinodular HCC despite three earlier rounds of transcatheter arterial chemoembolization (TACE). She had received a blood transfusion during a partial gastrectomy for a gastric ulcer at 36 years of age. There were no extrahepatic manifestations of hepatitis C at presentation, nor had she been taking any medications. Ammonia level was 60 μg/dL (normal, <70 μg/dL). Serum alpha fetoprotein and des-γ-carboxy prothrombin were 146 ng/mL and 1250 mAU/mL, respectively. A computed tomography (CT) scan revealed multiple carcinomas, including a hilus tumor of maximum 6.5 cm diameter with a portal vein tumor thrombus (Figure 1). Metastasis was not evident. Sorafenib at a daily dosage of 800mg was started in September 2009. After three days of treatment, the patient suddenly experienced disturbed consciousness and fell into a coma, which was suggestive of hepatic encephalopathy. Her serum ammonia level was high at 177 μg/dL (normal < 60 μg/dL). CT scan and magnetic resonance imaging of the brain showed no cerebrovascular disorder or reversible posterior leukoencephalopathy syndrome. [2] An electroencephalogram was not performed. Surprisingly, a CT scan of the abdomen showed a marked decrease in tumor enhancement, which indicated diminished blood flow (Figure 2). Sorafenib was discontinued at once, but the patient progressed to liver failure and died two weeks later.
| ||||||
| ||||||
| ||||||
|
Discussion
| ||||||
|
This case shows two critical implications of sorafenib administration for clinicians. First, the possible associations between hepatic encephalopathy and sorafenib have been reported, [3] [4]
[5]
and there have recently been several cases of serious adverse events leading to death. Furthermore, hepatic encephalopathy was reported in 34 of 2375 (1.4%) in Japanese patients in a blue letter by Bayer, which was higher than that in other countries. Thus, it should be investigated whether there are any differences in host factors or underlying liver diseases between Japanese and other ethnicities. Additionally, although sorafenib was administered for only three days, a clear decrease in tumor enhancement on the CT was evident. There have been no reports of such rapid changes in a patient with HCC treated with sorafenib to date. It has been suggested that sorafenib may exert beneficial effects through the inhibition of Raf serine/threonine kinases and tyrosine kinase receptors involved in tumor growth and angiogenesis. [6]
Sorafenib clearly had strong antiangiogenic effects in our patient, but its mechanism is not precisely known.
| ||||||
|
Conclusion
| ||||||
|
As hepatic encephalopathy is a life-threatening complication that may develop quickly, clinicians should be aware of this adverse event when using sorafenib for HCC, especially when it is concomitantly diagnosed with Child-Pugh A. In particular, when a rapid decrease in tumoral blood flow is evident on CT scan, patients should be followed carefully for this possibility.
| ||||||
|
References
| ||||||
| ||||||
| [HTML Abstract] [PDF Full Text] |
|
Author Contributions:
Author Contributions
Takefumi Kimura - Conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Final approval of the version to be published Takeji Umemura - Conception and design, Analysis and interpretation of data, Drafting the article, Final approval of the version to be published Tetsuya Ichijo - Analysis and interpretation of data, Critical revision of the article, Final approval of the version to be published Nobuyoshi Yamamura - Analysis and interpretation of data, Critical revision of the article, Final approval of the version to be published Eiji Tanaka - Analysis and interpretation of data, Critical revision of the article, Final approval of the version to be published |
|
Guarantor of submission:
The corresponding author is the guarantor of submission. |
|
Source of support:
None |
|
Conflict of interest:
The author(s) declare no conflict of interests |
|
Copyright:
© Takefumi Kimura et. al. 2010; This article is distributed the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any means provided the original authors and original publisher are properly credited. (Please see Copyright Policy for more information.) |
|
|